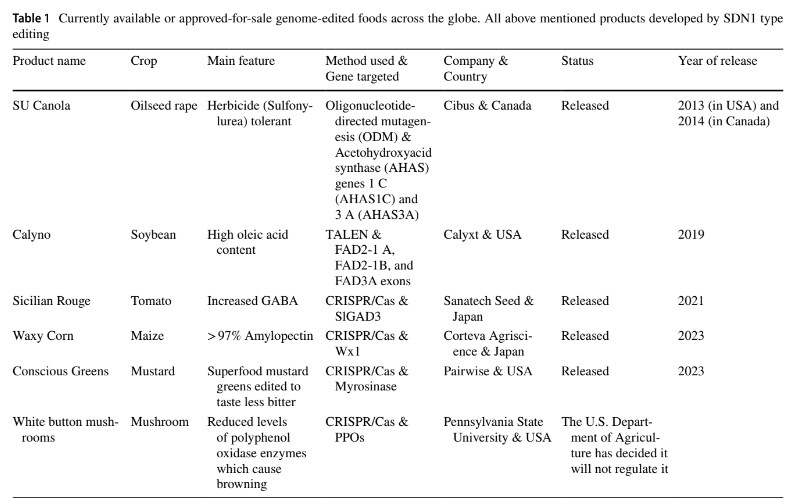
Despite the absence of foreign genetic material in crops developed through CRISPR/Cas mediated genome editing, there is an ongoing and intense debate surrounding the regulation of these crops prior to their release into the market. While certain CRISPR-edited crops have already been introduced in Japan, their legal status remains a point of contention in several nations, including the EU and New Zealand. This review paper serves as a comprehensive guide to the worldwide regulatory framework for CRISPR-edited crops, as well as provide insights into the future prospects of this transformative technology. By examining the current landscape of regulations and exploring potential avenues for harmonization, we can better understand the challenges and opportunities that lie ahead for CRISPR-edited crops.
Up until now, CRISPR crops have managed to steer clear of the societal stigma and outright legal bans that GMO crops have encountered. Many countries have different regulatory standards for biosafety laws. It’s essential to differentiate CRISPR technology from earlier GMO technology. The open and unbiased debate is needed among scientists, governments, business interests, consumers, and media In order to create a special global regulatory framework for genome-edited crops.