Several boys who were barely able to move and were completely dependent on ventilators are now eating, making sounds and walking with assistance, while the protein their bodies had been unable to manufacture is accumulating in their muscle cells. Audentes Therapeutics presented the results at a Presidential Symposium that highlighted several strides during the past year against rare neuromuscular diseases, including muscular dystrophies and spinal muscular atrophy. The interim data for MTM are here.

A dire diagnosis
The story of gene therapy for MTM begins with Alison Rockett Frase and Paul Frase, and their son Joshua. She was an assistant personnel manager for Guns’N’Roses, he a defensive lineman for the NFL. They’re what I call catalyst parents – moms and dads who refuse to listen to the refrain of “bring your baby home to die” and “there’s nothing we can do.” These parents push for progress, even if it comes too late for their own children.
Alison’s pregnancy was mostly uneventful, although an ultrasound scan at 22 weeks showed a fetal body a bit small for the head. That would turn out to be a hint.
By 38 weeks the baby-to-be had settled into a breech position, but when a physician tried to maneuver him into a head-down position, he’d flip back. Another clue?
When Joshua was born by C-section a few days later, on February 2, 1995, everyone in the delivery room could see that something was wrong. He was small, skinny, dusky and eerily silent.
Alison and Paul heard the word “floppy” to describe their baby’s absent muscle tone. Nurses began to prepare the young couple for their newborn’s imminent demise. But Joshua Frase had other ideas.
On the baby’s 24th day, Alison and Paul took him home and began the complex care required to help him to eat and breathe that would sustain him for nearly 16 years. Diagnosis came soon, a muscle biopsy revealing MTM, the mutation inherited from Alison’s X chromosome.
Joshua’s prognosis couldn’t have been much worse: 75 percent of boys with MTM die in weeks or months of respiratory failure, and the average life expectancy is 29 months. The weak muscles affect development of the skeletal system, and scoliosis and a large head result.
In MTM the protein myotubularin 1 is absent or abnormal, stemming from a missing or mutant MTM1 gene. Skeletal muscle cells remain in a fetal-like state.
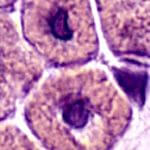
MTM is a “centronuclear myopathy,” which means that skeletal muscle cell nuclei are centrally located, rather than crowded to the sides of the abundant contractile proteins that fill the long, spindly cells. The unusual structure prevents calcium from entering the muscle cells and they can’t contract, like a cell phone unable to receive signals.
Assembling the team
Joshua Frase astounded his parents and caregivers with his will to live. But time was running out.
“As we approached his first birthday, a milestone we never truly expected to make, we began to realize that if we didn’t do something for our son, nobody else would,” Alison recalled.
That meant forming a research foundation, fundraising (helped by celebrity connections), assembling a team of investigators and learning the science. The Joshua Frase Foundation for Congenital Myopathy Research was born three weeks after the boy’s first birthday. Only 55 cases were known in the world then. MTM affects 1 in 50,000 live male births.
The road to gene therapy was long.
The team began to self-assemble in 1997, when Alison’s mother saw a story on CNN about Anthony Atala, the superstar of regenerative medicine who famously grew a human bladder in 8 weeks at Boston Children’s Hospital. Alison’s mom contacted him and he not only responded, but put together a dream team that included Alan Beggs and Louis Kunkel at Boston Children’s, Anna Buj Bello (who had a mouse model) from Genethon, Martin Childers and David Mack at the University of Washington, Jocelyn Laporte (who discovered the MTM1 gene) at the Institut de Génétique et de Biologie Moléculaire et Cellulaire in France, and others.
Over the years, as the Joshua Frase Foundation raised millions, the NIH provided funding too, as did other families.
By early 2014, gene therapy was working in mice and in dogs. In 2017 a report on body-wide delivery of the gene in dogs would prove critical to the early success of the gene therapy in boys.
“All readouts in the dog studies turned out to predict, with great accuracy, the outcome measures now being observed in the treated boys,” Dr. Mack said. For example, the dog work revealed that myotubularin levels of 10-40 percent are enough to restore muscle structure and function, which helped in determining dosages to test.
Dogs are the heroes and especially the heroines of the MTM gene therapy success story.
Labrador retrievers join the team
At a scientific meeting more than a decade ago, veterinarian Diane Shelton approached Dr. Beggs, who’d shown slides of MTM skeletal muscle in his presentation. Dr. Shelton mentioned that she had skeletal muscle samples from animals with “wasting puppy syndrome” from a veterinarian in Canada, and the cells with the central nuclei reminded her of the cells from the boys with MTM. Those dogs would turn out to have MTM; puppies typically didn’t survive beyond 5 months.
This was an important find: A natural dog model was critical in developing gene-based treatments for Duchenne muscular dystrophy and the first gene therapy approved in the U.S., to treat a form of blindness.

In 2008 Alison and Paul learned, through their network of experts, of Nibs, a female chocolate lab living happily on a farm in Saskatoon, Saskatchewan. Nibs was a carrier for a dog myotubularin mutation. The owners offered to give Nibs to the couple after Alison explained that the canine could be bred to found a colony that could be used to develop and test a gene therapy for the lethal neuromuscular disease.
So Alison flew out to collect Nibs, and settled her on a farm near Wake Forest University, headquarters of the gene therapy research. Alison told the astonishing tale in the journal Human Gene Therapy.
The very special chocolate lab had pups and the gene therapy begun on her descendants in 2013 worked. This paper assesses two of the treated dogs, Pavlov and Turing. Four years post-gene therapy, they had normal neuromuscular function, suggesting the disease could be stopped and maybe even reversed or prevented. Pavlov and Turing turned 7 on May 1, the day that Audentes announced the good news about the treated boys at the gene therapy meeting. The dogs are still doing well.
The gene therapy worked so well that one dog, Rocky, instead of dying by 5 months, fathered a litter with a female carrier of MTM. That mating begat females who had a double dose of the mutation. Affected females, healthy because of the treatment, wouldn’t exist in nature because a male dog couldn’t survive long enough to mate to produce them. (Females with MTM have mutations on both X chromosomes.)
With more dogs available, the clinical trial could be planned. Several dogs were required because the treated animals only have the gene therapy in their muscle cells, not sperm or eggs, so they can’t transmit the disease correction. And gene therapy adds a gene, it doesn’t replace the mutant one.
The clinical trial
In summer 2012, Paul Frase met with entrepreneur Matt Patterson, who would become CEO of Audentes Therapeutics, and presented the preclinical data that the Joshua Frase Foundation had funded.
 Planning the gene therapy had three stages:
Planning the gene therapy had three stages:
RECENSUS reviewed medical records for 112 boys “so we could learn about the disease other than from the few publications,” Patterson said. The records showed mortality in 64 percent of patients before 18 months and in 32 percent of patients older than 18 months.
INCEPTUS conducts a clinical assessment of MTM before treatment, which is useful in evaluating response to gene therapy for trial participants.
ASPIRO is the phase 1/2 clinical trial begun in 2017.
Early results
The treatment, AT132, is delivered intravenously to an arm or leg in an adeno-associated virus (AAV) 8 vector. Eleven boys under age 6 have received the treatment, at UCLA Medical Center, the University of Florida, and the Ann & Robert H. Lurie Children’s Hospital of Chicago.
Seven boys got 100 trillion viruses per kilogram of body weight and four received triple that dose. The boys at the higher dose now make more of the missing protein than do healthy children.
Signs of success appeared at 12 weeks, according to a news release from Audentes issued in 2018. The company presented even better news for the 24-week mark for 7 patients at the American Society of Gene and Cell Therapy annual meeting last year.

Today, all the boys are meeting motor milestones and are less dependent on mechanical ventilation, with four boys off it completely. Being vent-free means it’s possible to survive, to live with the disease that claimed Joshua Frase. Muscle biopsies show normal function in the boys and the safety profile is good.
A year ago Alison saw the videos of the first treated boys lifting their arms and reaching for balls and was astonished. “Watching this unfold before my eyes has been almost surreal. Am I really witnessing my son’s dream? The clinical trail is bringing 22 years of painstaking work to real tangible results some would consider miraculous,” she said.
It will still take time to see if effects are sustained, and whether symptoms never seen before emerge because boys with the disease live longer. But for now, gene therapy for MTM is on the trajectory towards regulatory approval. And that’s great news.
It will be too late for Joshua Frase, who died in 2010. But it will be his legacy.
Thanks to the Frase family for use of the photos.
Ricki Lewis is the GLP’s senior contributing writer focusing on gene therapy and gene editing. She has a PhD in genetics and is a genetic counselor, science writer and author of The Forever Fix: Gene Therapy and the Boy Who Saved It, the only popular book about gene therapy. BIO. Follow her at her website or Twitter @rickilewis































