The commissioner has the authority to allow unapproved medical products to be used in an emergency when there are no adequate or approved alternatives. An EUA is not the same as full approval and it can be withdrawn.
…
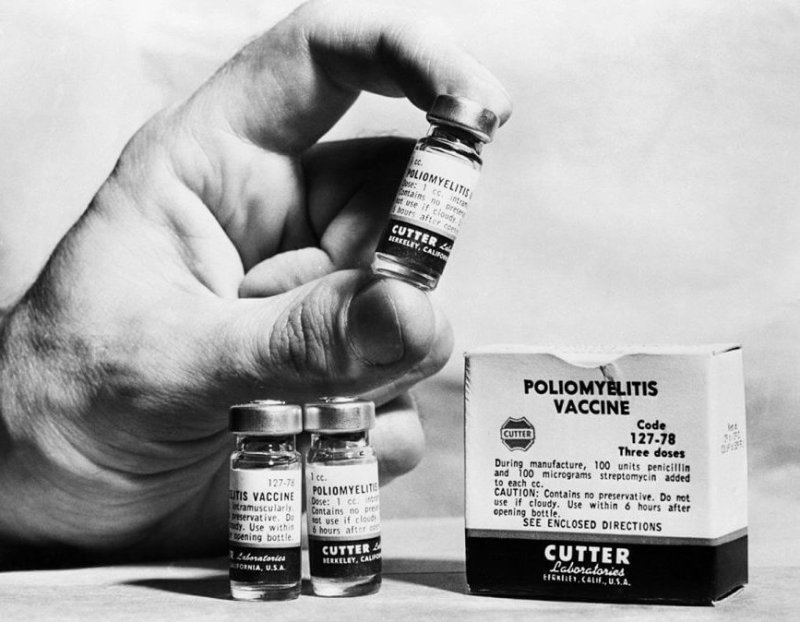
[In 1955,] more than 200,000 children got the polio vaccine, but within days the government had to abandon the program.“Forty thousand kids got polio. Some had low levels, a couple hundred were left with paralysis, and about 10 died,” said Dr. Howard Markel, a pediatrician.
Markel said people’s mistrust of the system makes the idea that the FDA would rush this process before late stage clinical trials are complete “colossally stupid.”
“This is one of the most ridiculous things I’ve heard this administration say,” Markel said. “All it takes is one bad side effect to basically botch a vaccine program that we desperately need against this virus. It’s a prescription for disaster.”
FDA Commissioner Hahn said that the vaccine decision will be based on data, not politics.