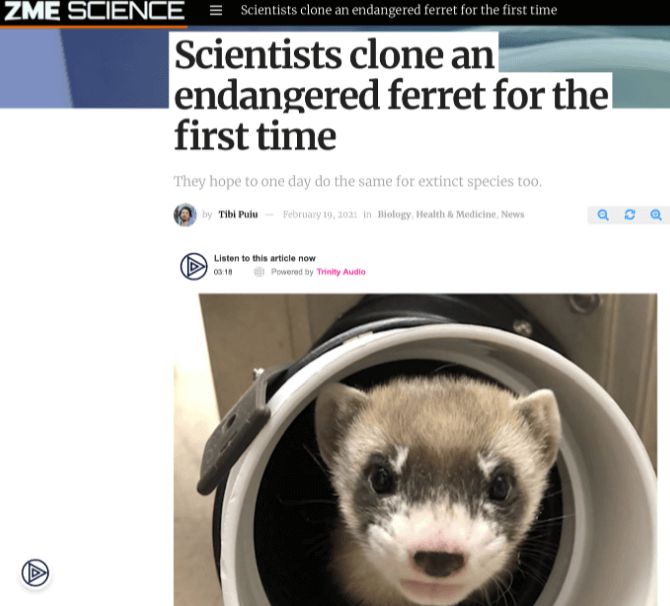
From Borneo to Britain, it’s the scientific breakthrough that captured the world’s attention. No, not the Perseverance rover landing on Mars, but the story of Elizabeth Ann, the ever-so-cute baby black-footed ferret cloned from the cells of a long-deceased female (cryopreserved at the San Diego Frozen Zoo).
It seems an inspiring story of “bringing biotechnology to conservation,”, to quote the mission statement of project leader Revive & Restore, whose aim is “to enhance biodiversity through the genetic rescue of endangered and extinct species”. Yet, like many scientific advances, Elizabeth Ann’s birth raises questions.
Future of ferrets—and of cloning
Once widespread across the American West, black-footed ferret numbers had plummeted to near extinction by the late 20th century. Today, all remaining members of the species, the continent’s only native ferret, are descended from seven individuals from a single colony discovered in 1981. All, that is, except for the latest arrival, Elizabeth Ann, whose future offspring (so it’s hoped) will reintroduce much-needed genetic diversity into the inbred ferret population.
So far, the success of the project has addressed a number of common conservation concerns about the use of biotechnology. For a start, and despite the extreme demands from some quarters that genetic manipulation of any kind should be banned, Elizabeth Ann demonstrates that cloning and other genetic techniques can indeed play a positive part in wildlife conservation efforts.
Admittedly, this follows a number of less successful conservation cloning experiments. Attempts in 2003 to clone the wild Javan banteng, an endangered species of cattle resulted in only two full-term calves; one was euthanised shortly after birth, the other survived but lived only half the expected banteng lifespan. In the same year, efforts to clone the recently-extinct bucardo or Pyrenean ibex suffered a similar string of setbacks: of the 208 cloned embryos implanted in surrogate goats, just a single calf made it to term, only to die painfully of respiratory failure within minutes of birth.

The brief existence of the unfortunate bucardo calf marked the initial proof of concept of de-extinction, of (eventually) using biotechnology to resurrect the likes of the woolly mammoth or the passenger pigeon. This controversial idea highlights another forceful criticism of bioengineering in conservation, namely the ‘moral hazard’ fear that the ability to revive species will reduce the incentive to protect endangered wildlife in the here and now. If extinction is not forever and if we can always ‘bring them back’ tomorrow, why bother saving them today?
This point was forcefully made by University of California, Santa Barbara ecologist Douglas McCauley in a debate over de-extinction:
“Honestly, the thing that scares me most is that the public absorbs the misimpression that extinction is no longer scary. That the mindset becomes: Deforest, no biggie, we can reforest. If we drive something extinct, no biggie, we can de-extinct it,” he’s said.”
Space precludes further discussion of the pros and cons of de-extinction (though see here and here for two sides of the debate). Suffice to say, the black-footed ferret experiment shows that advanced ‘genetic rescue’ need not induce moral hazard complacency.
Short-tailed weasels and unintended consequences
Paradoxically, the possible use of biotechnology by conservationists in New Zealand could intentionally lead to an opposite outcome of the black-footed ferret experiment —namely the complete eradication of a related mustelid species, the short-tailed weasel or stoat (part of the family of carnivorous mammals that includes ferrets, badgers and otters). The stoat example illustrates by far the greatest concern over the use of bioengineering in conservation contexts: it’s potential for environmentally devastating unintended outcomes.
Introduced in the 19th century to control rabbits (yet another misguided introduction), stoats have since become “public enemy number one” for New Zealand’s native birds, decimating the lingering populations of some of the world’s rarest species. (The Department of Conversation’s no-nonsense description of stoats states they are “voracious and relentless hunters … having only two reasons for living—to eat and to reproduce”.) In a further irony, another endangered species sponsored by Revive & Restore, the New Zealand takahē, is under immense threat from Elizabeth Ann’s close relative, the stoat.

Given the massive menace of these mustelids, New Zealand scientists have recently sequenced the stoat genome in an effort to develop targeted poisons. In addition, in light of the ambitious goal to rid the country of all introduced predators by 2050, the use of bio-engineered ‘gene drives’ (suites of genes that cause population decline within a specific species) have also been mooted. Other introduced pests, such as rats, possums and wasps, are additional targets for such technologies.
Some environmentalists, including New Zealand’s influential Green Party, oppose the use of genetic engineering, even for seemingly laudable conservation causes (as indeed they reject using GE to help tackle climate change). Nevertheless, in the case of gene drives, at least, the Greens’ fear of the unintended consequences is at least understandable, especially in New Zealand, considering past experience. When rabbits were introduced as a food and game species they rapidly became an agricultural pest; when stoats were introduced to eradicate the rabbits, they turned on the native fauna instead.
While stoats, rats and wasps are invasive pests in New Zealand, they are important features of natural eco-systems elsewhere; possums, meanwhile, are a protected native species in their homeland, Australia. With respect to gene drive technology, therefore, the worst case scenario posits bio-engineered organisms escaping from the targeted area or country and driving species to global extinction. While such apocalyptic outcomes are perhaps overblown, conservation-minded geneticists are already designinging fail-safe mechanisms such as ‘daisy chains’ to limit the persistence of gene drives within a population.
Diversity—all it’s cracked up to be?
Another pertinent conservation issue — exemplified by both black-footed ferrets and the environmental situation in New Zealand—are the widely-used concepts of (interspecific) biodiversity and (intraspecific) genetic variation. The orthodox view within conservation biology is the more diversity and variation the better.
This, though, is not always the case. A recent PNAS paper, for example, argues strongly against the “fundamental assumption … that higher levels of genetic diversity lead to an increase in fitness and long-term survival of a species”. The authors claim that, while lack of genetic variation may be a contributing factor in population decline, “supporting empirical evidence for the existence of a causal relationship between genetic diversity and population viability or adaptive potential is weak”. Overall genetic diversity within a species is a crude measure of long-term viability because much of that diversity is ‘neutral’, neither enhancing nor diminishing survival or reproduction.
Our own species provides relevant examples, such as “the Out-of-Africa bottleneck ∼50,000y ago [that] strongly reduced neutral genetic diversity in non-African human populations”. Yet, as well as losing broader genetic diversity during this bottleneck, many of the more deleterious mutations were also lost. Thus the Greenland Inuit, “one of the most extremely bottlenecked human populations” in the world, actually carry fewer deleterious mutations overall than more genetically diverse populations.

A further example of such bottlenecking not discussed in the PNAS paper is the dispersal of terrestrial fauna to mid-ocean islands through random accident—storm-blown birds, say, or animals on driftwood swept out to sea. Given some of the distances involved and the chance nature of such events, thriving later populations of some species could conceivably have descended from a single pregnant female.
As an apt example, New Zealand’s rails or ‘swamphens’, including the flightless takahē are likely wind-blown arrivals from Australia, over 1000 kilometres away. Archaeological evidence suggests the most recent of these rails, the pūkeko or purple swamphen, arrived only within the past 300 years (perhaps filling a niche formerly occupied by the takahē); despite its extreme bottlenecked origin, this species has successfully spread throughout the country.
In short, the recent rapid decline and/or extinction of island species is not the result of limited genetic diversity; rather, the immediate cause is the same as that still threatening most endangered species elsewhere—habitat destruction. As the authors of the PNAS paper emphasise, fixating on genetic diversity can shift attention away from the larger problem: “Nongenetic factors such as overkill, habitat destruction and fragmentation, the introduction of invasive species, rapid climate change, or pollution, reduce the growth rates of species and cause their populations to decline.”
This is not to say that measures of genetic variation are misguided in conservation, rather, as the PNAS paper notes, “Researchers need to investigate for each species individually which genetic mutations allow the species to thrive and which mutations lead to diseases that can threaten the species. There is certainly no simple ‘one-size-fits-all’ measure of extinction risk”. The take home message is that genetic technology is a useful tool in conservation efforts, not a silver bullet.
The ‘natural’ environment?
What about the broader concept of biodiversity? The notion of biodiversity is popularly synonymous with healthy, functional ecosystems. This is misguided. As a result of human expansion, most modern environments are now far more biodiverse than they were in the past. Yet this masks the threat to existing ecosystems from non-native organisms, from trivial-seeming examples like American grey squirrels in Britain to more worrisome Asian giant hornets in the US to the multiple pest species in New Zealand.
The lay conception of biodiversity is often over-romanticized as a pristine state of nature, and human-induced environmental change is often regarded as inherently bad. But is it? Consideer Ascension Island in the Atlantic; “once a barren volcanic rock, [the island] is now much greener thanks to a deliberate policy, suggested by Charles Darwin, of bringing in plants from elsewhere in the tropics to create a forest ecosystem”. Now wonderfully biodiverse, Ascension’s ‘natural state’ has nevertheless been deliberated altered. Is that a good thing or a bad thing?
Or take the laudable efforts to return some of the lost biodiversity of the US, epitomized by the cloning of Elizabeth Ann. North America’s natural state is often taken to be that existing before 1492. But why choose that specific date? Loss of biodiversity (most especially of native megafauna) predates European colonisation by many thousands of years—hence arguments for the ‘Pleistocene rewilding’ of the continent; deliberately introducing proxies for extinct fauna (e.g. lions for sabretooth cats, elephants for mastodons) to recreate long-degraded natural ecosystems. Similar reasoning, of attempting to restore valuable pre-existing ecosystems, lies behind the concept of de-extinction discussed above. Not surprisingly, Revive & Restore is at the forefront of the de-extinction movement.
Conservation biologist Philip Seddon, lead author of the International Union for Conservation of Nature’s guidelines for de-extinction research, acknowledged much of the wider concern over the use of genetic biotechnology within the conservation community:
“The spread of genes can be difficult to control. We probably won’t lose track of mammoths in Siberia, but what about rats? It becomes hard to control those sorts of populations. And there are the same fears one might have about genetically modified crops—the idea that a modification may move into relatives, may jump in and out, or may not be expressed in the way that you expect.”
Human values versus valuable science
Seddon’s approach to biotechnology and conservation , however, is reflective not reflexive. He emphasized the ‘clash of cultures’ between conservation biologists and synthetic biologists, with “little overlap in the two communities’ respective training, fields of practice, and views of the future”.
“Conservation practitioners and conservation biologists are, in general, conservative and precautionary. Their focus is on preservation and restoration, limiting human impacts, and slowing or halting biodiversity declines. They have been characterized as looking to the past and being risk averse. … What is the way forward? Can we engage a skeptical and cautious conservation community, harness the enthusiasm of the public imagination, and ensure that risks are understood and managed?”
A possible way past this impasse, Seddon suggested, “is through cautious application of synthetic biology” in familiar conservation contexts: reintroducing lost genetic diversity, say, or controlling wildlife diseases.
Early and demonstrable success … [with] these types of intractable conservation challenges will do much to start a more informed debate about human management and manipulation of the natural world.
And this brings us full-circle, back to a cloned baby ferret, whose birth so brilliantly blends conservation, biotech and public imagination. By its very nature, conservation is driven by subjective human values not objective scientific facts. Science can tell us about the past and current state of an environment, and help predict what will happen in the future. And modern genetic technology has the potential to massively increase our abilities to monitor and alter environments in ways that further our conservation goals. But science alone cannot tell us what we should or shouldn’t do in our interactions with nature.
With this in mind, we should consider Revive & Restore’s call to expand “the narrative of conservation” away from the “fear of unintended consequences [that] still paralyzes conservation innovation”. Instead, “genetics-based advancements,” alongside traditional conservation practices and values, could bring the possibility for intentional change—for intended consequences—within our grasp. This is not to blindly endorse that goal, any more than it is accepting genetic technology as the cure-all for our current conservation woes. Yet, with the ecological stakes so high, at the very least it’s an idea worth debating.
Patrick Whittle has a PhD in philosophy and is a New Zealand-based freelance writer with a particular interest in the social and political implications of biological science. Follow him on his website patrickmichaelwhittle.com or on Twitter @WhittlePM