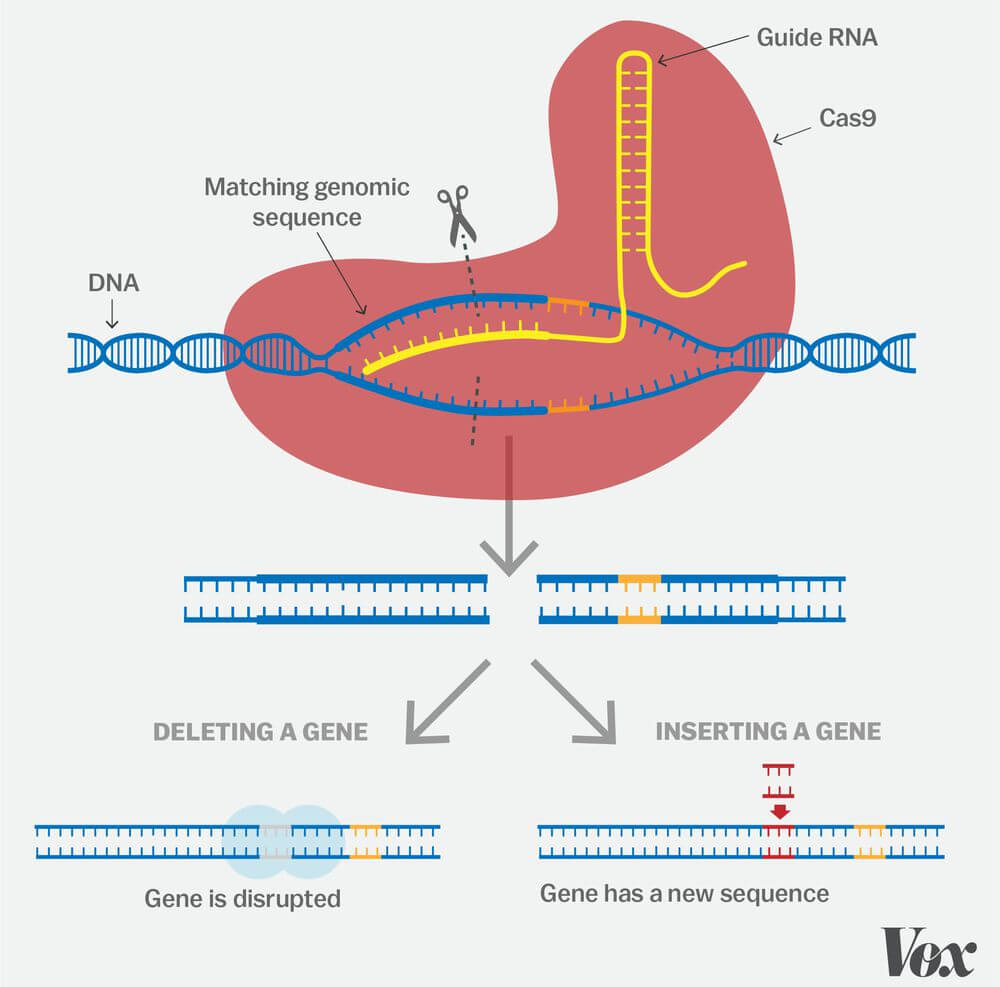
Gene Editing / CRISPR
Can promises of gene drive technologies overcome safety concerns?
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. Gene drive enthusiasts say ...
CRISPR-based gene therapy may provide cure to Duchenne muscular dystrophy
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. In an amazing, salutary ...
How did ‘CRISPR’ get its name?
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. A new gene-editing technology ...

Why the concept of GMOs is meaningless
European Union legislation on agricultural genetics and biotechnologies is hopelessly warped — and it threatens to get worse with the introduction ...
CRISPR’s promise may expand beyond genome editing
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. Three years ago, Stanley ...

Gene therapy is doing all that we’d hoped for* *(so far)
New gene therapies are showing evidence of advancing our treatment of genetic diseases. Scientists are 5 years along the 15 ...
CRISPR patent dispute to be decided on legal technicality
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. The U.S. Patent Office ...
Gene drive technique aims to control mosquito, pest populations
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. Biologists in the United ...
Caution needed as genetic testing comes to workplace
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. According to a recent ...
CRISPR technology draws mounting interest from investors
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. In a single year, ...
Human genetic modification may promise cures for some diseases
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. “Human gene therapy” has ...

Gene editing propels search for Duchenne muscular dystrophy cure
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. After decades of disappointingly ...

Scientist who developed CRISPR reflects on year of ethical debate
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. Some 20 months ago, ...
Australian, New Zealand food regulators soon to decide if gene-edited crops are GMOs
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. Australia's food regulator, Food ...
CRISPR edited bacteria hold great potential for cultured foods, probiotics, list goes on
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. The on-going CRISPR craze ...
CRISPR named Science’s 2015 Breakthrough of the Year
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. It was conceived after ...
Gene editing protects pigs from untreatable virus, without adding foreign DNA
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. This latest [genetic engineering] ...
European researchers in limbo waiting for EU’s decision on whether CRISPR plants are GMOs
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. Plant geneticist Stefan Jansson ...
CRISPR lets scientists rewrite organisms’ body plan
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. The simplicity of CRISPR-Cas9 ...
Gene edited crops present PR challenge, innovation opportunity
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. Recent advances in the ...
CRISPR crops could help solve hunger, climate change challenges if they avoid PR mistakes of GMOs
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. As Newsweek pointed out ...

Scientists can engineer mosquitoes to render them sterile, crashing malaria carrying population
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. And now there are ...
Gene-edited pigs resistant to deadly virus may be ‘game-changer’ for pork producers
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. Since the Porcine Reproductive ...
White privilege? Will Western activists block CRISPR solution to protecting millions of Africans against malaria?
Scientists now have the ability to eradicate one the world's most deadly diseases--malaria, which kills as many as 2.7 million ...
Gene editing regulations can’t be decided by scientists alone
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. An international summit on ...
Will human genome editing beckon a new eugenics movement?
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. The technology is called ...

DIY gene editing at home? ‘Farma’ kits can produce home-brewed pharmaceuticals
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. MIT Media Lab graduate Will ...