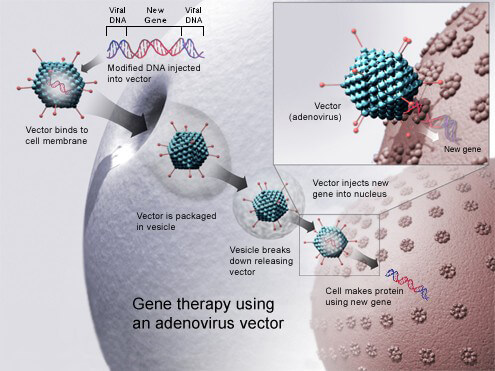
Gene Therapy
Gene therapy study produces promising results for hemophilia B patients
A fraction of patients with a common form of the bleeding disorder hemophilia develop anallergic reaction to the blood-clotting treatment they need to ...

Having one baby to save another
A couple plan to have specialized IVF so they can have a second baby and use umbilical cord blood to ...
Gene therapy with a difference
Terri Ellsworth is convinced that her 12-year-old son Billy, who has Duchenne muscular dystrophy, is being helped by an experimental drug ...
Defanged HIV a positive for gene therapy
The scientific community witnessed an unexpected medical breakthrough last year, when a child with acute lymphoblastic leukemia (ALL) was treated ...
The science and troubling ethics of gene therapy
Why is progress on gene therapy—the treatment of genetic disorders by giving sick people doses of the healthy genes they ...
Heart gene therapy trial begins
It is 18 months since Carol Gedda suffered a massive heart attack. It left her with just 20% of her ...
Heart attack damage repaired using gene therapy
Heart attacks cause cells in the affected area to stop beating and become encased in scar tissue, but researchers believe ...
Bioengineering modular viruses to fight disease
Imagine fitting viruses together like Lego pieces and using them as a vehicle to deliver gene therapy. Scientists at Rice ...
The fall and rise of gene therapy
Rarely does a whole life’s work crumble in a single week, but James Wilson’s did. Wilson and his colleagues were ...
Gene therapy used to improve memory in mice
Researchers for years have been on a tantalizing quest to develop a drug to slow age-related memory loss. In recent research, ...
How gene therapy helped one bubble boy
Two-year-old Jameson Golliday was born with X-SCID, or "bubble boy disease," which means he has no immune system. At birth he had ...
Would you edit your genes?
The most difficult thinking about gene therapy involves normally healthy people who have mutations that are highly predictive of a ...
Spliceosome may help disable foreign RNA from viral infections
Although small interfering RNAs (siRNAs) are largely explored today for their potential in gene therapy, the phenomenon was first described ...
NIH begins gene therapy trial for Parkinson’s disease
All eyes were on Perry Cohen when he froze at the microphone. His voice failed him. He couldn’t read his ...
UK scientists develop brain disease therapy
UK scientists have developed a new stem cell therapy that could, in the best case scenario, offer a "largely curative" ...
Researchers bioengineer a new technique for improving gene therapy results
Gene therapy researchers have produced a bioengineered decoy that fools the immune system and prevents it from mistakenly defeating the ...
Gene therapy for baby born without immune system
Doctors re-engineered a girl's DNA using a ‘reprogrammed virus’ to splice in the vital gene she is missing into her ...
Testing of gene therapies expanding at Boston-area hospitals
Years after it was hyped, vilified for the death of a teenager, and then mostly forgotten by the general public, ...
Is gene therapy coming of age?
Researchers are promising results from gene therapy trials for the treatment of two different rare diseases ...
Gene therapy using HIV helps children with fatal diseases, study says
Italian researchers have used a defanged version of HIV to replace faulty genes — and eliminate devastating symptoms — in children suffering ...
DNA screening is part of the new eugenics—and that’s okay
Modern genetic screening is not driven by a draconian desire to “improve” the species. This "new eugenics" is driven by ...
Gene therapy cures severe pediatric neurodegenerative disease in animal models
The following is an excerpt. A single session of a gene therapy developed by the Universitat Autònoma de Barcelona (UAB) ...
DNA transfer in human eggs prevents disease
The following is an excerpt. Scientists have for the first time transferred DNA between human eggs. The success at Newcastle ...
Scientists create gene therapy virus that targets the retina
The following is an excerpt. Using mouse eyes as a setting for directed evolution, scientists have created a new version ...
Scientists restore long-term memory to mice
The following is an excerpt. University of California-Irvine neurobiologists have discovered a protein complex in neurons that is essential to ...
Gene therapy helps muscular dystrophy patients breathe easier
The following is an excerpt. Children with a rare form of muscular dystrophy called Pompe disease often spend their days ...
Gene delivery via magnetic nanoparticles
The following is an excerpt. Stent angioplasty saves lives, but there often are side effects and complications related to the ...