The television program 60 Minutes spent nearly an entire hour Sunday covering a new treatment for brain cancer. Using words like ‘extremely promising’ and ‘cure,’ the show was so overly positive that it was easy to miss the fine print: half the patients in the trial were already dead and only three had cancers in remission, the goal of treatment.
When the media covers medical breakthroughs, especially those centered around genetics, there are several levels of hype that need to be considered: cost, number of people who could use it, the treatment’s staging in the federal approval process and the other treatments available and those under study for the same condition. At best, about one out of 100 treatments under development at any time will be useful. And those patients who read about the other 99 without diligent reporting may have false hope about their health and expected number of remaining years. Or worse, they may try to access untested treatments outside of clinical trials and cause themselves harm.
In the clinical trial profiled in the 60 Minutes piece, Duke University researchers genetically modified the polio virus to make it deadly to cancer cells but unable to infect healthy cells in the body. The modified polio virus can only divide within tumor cells. This essentially coats the tumor with a virus our immune system knows to fight. The immune system takes over and kills the tumor cells. The polio part, while sexy, doesn’t really matter. Similar experiments have been done with herpes and cowpox viruses by other groups working on cancer killing immunotherapies reports Arlene Weintraub at Forbes.
Other cancer-fighting immunotherapies rev up patient’s own T-cells, a type of white blood cell, to fight blood cancers. Those results were similarly promising: about half of patients had some positive results.
So, while it may be fair to say that immunotherapy for cancers is a promising new area of research, to call out any of these individual trials as a sure thing is a mistake. “There are… reasons to be wildly optimistic about the state of cancer research. One is that new treatments that harness the immune system to attack tumors do lead, in some cases, to long-lasting remissions, even cures,” Matthew Herper wrote at Forbes about hyping treatments still under development.
In 2003, researchers writing in the American Journal of Medicine discovered something that should change how you think about medical news. They looked at 101 studies published in top scientific journals between 1979 and 1983 that claimed a new therapy or medical technology was very promising. Only five, they found out, made it to market within a decade. Only one (ACE inhibitors, a pharmaceutical drug) was still extensively used at the time of their publication.
Media coverage of treatment breakthroughs is often flawed for many reasons. One of which, as Belluz points out is novelty. The ‘news’ is often based on new studies, techniques, chemicals, etc. Those results get reported simply because they are novel. But science works almost oppositely. A result should be vetted, replicated and picked apart before its accepted in the research dogma. But no one wants to report a study that found second-hand smoke to be carcinogenic again. That’s not news from the media’s perspective.
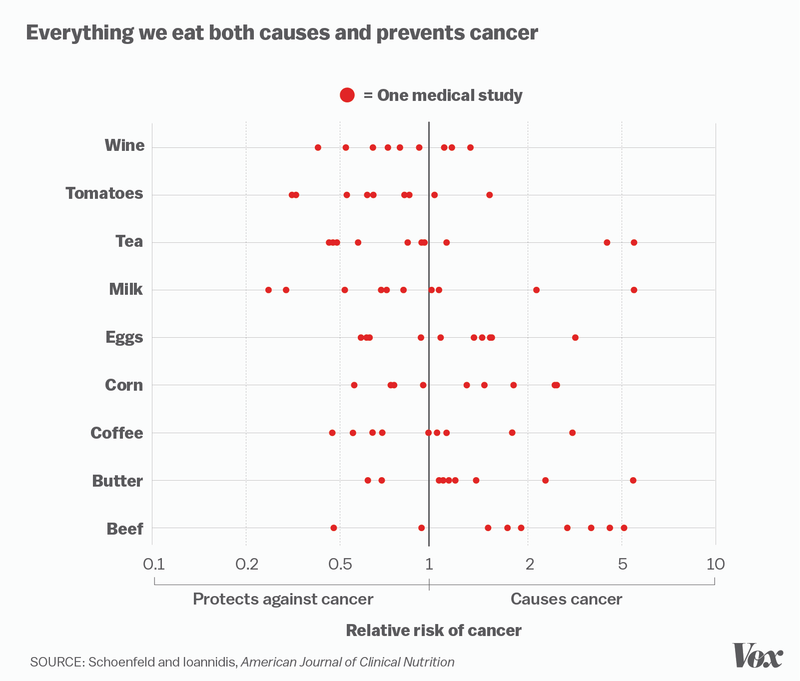
Nor is the back-and-forth of scientific findings. Belluz points to this graph showing the number of studies that indicate a certain food either causes or is protective of cancer. As it turns out, the effects aren’t clear at all. Looking at one study would lead to the incorrect conclusion.
Even if the evidence is certain and treatments work well, it can take more than a decade for a compound to make it out of a laboratory to the market as an FDA approved drug. The FDA, however, suggests it can be done in as little as 10 months.
The juxtaposition between the fast pace of the news cycle and the slow pace of science isn’t entirely to blame. Researchers are incentivized to publicize their work. Often, a scientist might have a financial stake in his or her work and money to be made popularizing a treatment under development. Additionally, institutions gain in prestige and income when more patients come to them for treatment. Even publicizing a disease might change funding allocations, freeing up more grant money for researchers focused on certain conditions to compete over.
In the 60 Minutes piece, for example, Matthias Gromeier, the microbiologist who helped design the immunotherapy therapy used at Duke lists a slew of cancers that his technique works on. “So we have done this for lung cancers, breast cancers, colorectal cancers, prostate cancers, pancreatic cancers, liver cancers, renal cancers. We probably see this in just about any type of cancer you can imagine,” Gromeier said.
But in reality, the treatment has only been tested on patients with one kind of brain cancer called glioblastoma. For rest of that list, ‘treatment’ happened in a petri dish in the lab writes Tom Watkins at Health News Review. The site is dedicated to vetting media reports about medical news.
Gromeier’s quote is not inaccurate but it is a bit misleading. Without specifically mentioning that treatment of these cancers are still in the basic research phase, thousands of people with these cancers may call doctors offices asking when they can get the polio-based treatment. And trials, especially early in the approval process are prohibitively small. There is no way to accommodate all the patients who want to be included.
In some circumstances, patients with severe disease may seek opportunities for cutting-edge treatments outside of the clinical trial system offered by unethical practitioners. People undergoing do-it-yourself fecal matter transplants are all over the internet. The treatments are gaining traction for people who suffer from bowel disease and resistant bacterial infections, but can be dangerous if parasites and other pathogens aren’t professionally removed from the donor stool.
Dubious stem cell clinics in the U.S. and abroad advertise cures for ALS (Lou Gehrig’s disease), heart disease, multiple sclerosis and other conditions despite the fact that these treatments are, at best, early in their development. Some of these treatments aren’t even in trials yet. And stem cells carry risks of developing tumors and other cancers. Last year a woman who had participated in a stem cell trial for paralysis developed a tumor from the ultimately unsuccessful treatment.
Meredith Knight is contributor to the human genetics section for Genetic Literacy Project and a freelance science and health writer in Austin, Texas. Follow her @meremereknight.
Additional Resources:
- Buy your telomere testing kit here! Evidence based or psuedo-science? Genetic Literacy Project
- Cancer, bad luck and some lessons in science reporting, Genetic Literacy Project
- Why ‘precision medicine” initiative will fall short of hype, New York Times




