One of the principal hallmarks of the next generation of medicine is being able to take advantage of precision medicine, which simply stated is the ability to precisely target particular disease targets within the body without as many off-target effects (which lead to more side effects) as currently-available therapies.
The background of this research is incredibly interdisciplinary: for example, it tends to be technological advancements through engineering solutions which allow for clinicians to apply certain targeted therapies and the outcomes must still be measured through randomized controlled trials to ensure that the effects seen are not confused with natural variability or chance if they are to be taken seriously as contenders for new disease treatments.
These therapies represent the interface of biochemistry, clinical research, engineering, medical practice, pharmacology, and even other disciplines. An example of such a technology was the gastric drug delivery device (payload delivery device), which I previously described here.
Turning to the simple to solve the most complex

Some new research explores the power of continued progress in this field: Scientists from the United States, Australia and Germany have executed trials using a genetically engineered algae to deliver chemotherapeutic drugs selectively to cancerous cells, which killed about 90 percent of them in human cell culture.
Diatom algae are a major type of photosynthesizing microalgae whose exterior skeleton (called a frustule) is comprised largely of silica and water (hydrated silica) – structurally developed by the organism itself, and is thusly called biosilica. Publishing their study in the journal Nature Communications, the team also found that when they injected the nanoparticles into mice, tumors regressed.
“Although it is still early days, this novel drug delivery system based on a biotechnologically tailored, renewable material holds a lot of potential for the therapy of solid tumours including currently untreatable brain tumours,” said Nico Voelcker, from the University of South Australia, who partnered with researchers in Dresden, Germany.
This study also highlights a major advance in the use of nanoparticles in drug therapy. As an article in I Love Science noted:
The application of nanoparticles in this field has seen an uptick over the last decade. Often made of graphene – a genuinely multipurpose wonder material – these biodegradable-yet-tough carbon particles can be designed to imprison anti-cancer drugs inside them, transporting them through the bloodstream towards their target tumor. Some are even able to heat up when triggered, causing them to disintegrate their target cancer cells.
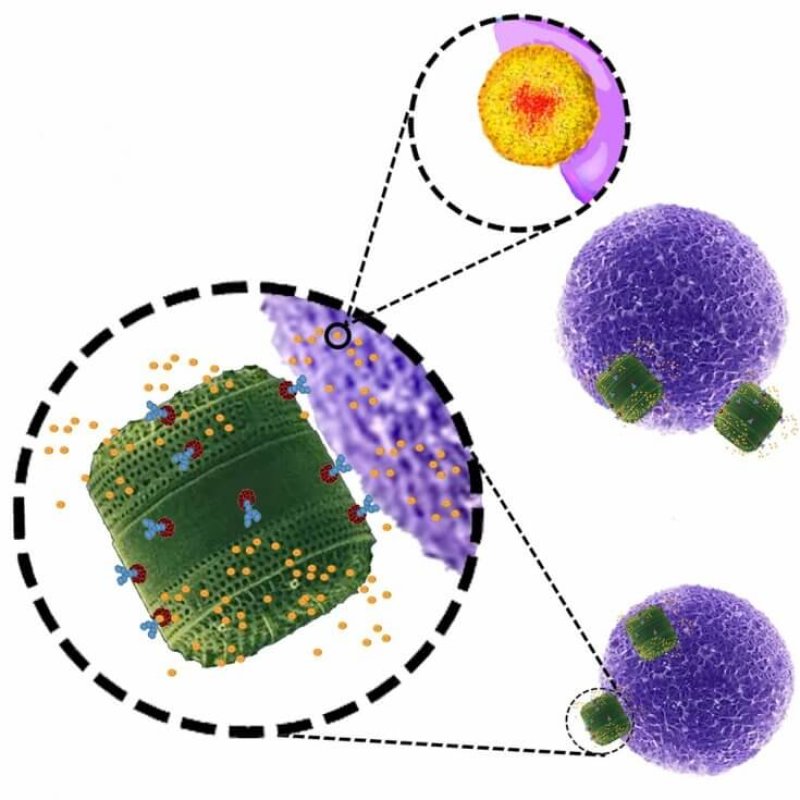
This new study takes the nanoparticle delivery method and modifies it slightly. Instead of using carbon-based nanoparticles, the researchers switched to silicon. In this instance, they used diatoms, a common type of photosynthesizing algae. These tiny organisms have silica-based skeletons that, although resistant to erosion, are ultimately biodegradable.
The algae Thalassiosira pseudonana was genetically modified to have an antibody binding domain (protein) on its exoskeleton surface, where the cancer treatment drug was attached. Why was this particular algae selected? Because, among other reasons, Thalassiosira was the first marine phytoplankton to have its entire genome sequenced over a decade ago.

The clever use of the diatoms takes advantage of some of the workarounds needed at the nanoscale, especially structural homogeneity. As Feynman observed (1959):
The metals that we use have a grain structure, and this would be very annoying at small scale because the material is not homogeneous. Plastics and glass and things of this amorphous nature are very much more homogeneous, and so we would have to make our machines out of such materials.
The biosilica shells of the algae have nanoporosity — regions of porousness where the morphology is on the nanoscale. This allowed the researchers to target neuroblastoma and B-lymphoma cells with the therapy drug in nanoparticle form — meaning the drug delivery nanosachet preferentially attaches to molecules only found on cancerous cells. In this way, it is an extension of the technology of antibody drug conjugates (ADCs). This allows a few benefits, including the ability to have the diatom biosilica selectively target the diseased cells, as well as to be able to deliver the anti-cancer drug directly to the target, which is normally a challenge because many of these drugs are poorly-soluble in water. Additionally, current nanoparticle manufacturing is extraordinarily specialized and is very expensive; Whereas to grow these modified algae requires very few process inputs beyond water, ultraviolet light, and carbon dioxide.
Beyond demonstrating the the drug can be effectively deployed on-target with minimal variation is one thing; Before any credible studies could be set up for human candidates, the safety of the diatoms in vivo would have to be rigorously studied for this use to show that the risk:benefit ratio clearly favored moving forward with further study.
Ben Locwin, Ph.D., M.B.A., M.S. is a contributor to the Genetic Literacy Project and is an author of a wide variety of scientific articles for books and magazines. He is also an expert contact for the American Association for the Advancement of Science (AAAS) and consultant for many industries including biological sciences, pharmaceutical, psychological, and academic. Follow him at @BenLocwin.