Are we trapped by our biological clocks? How much does age define us?
Famed personal trainer Jack LaLane seemed to be able to turn back the hands of time, maintaining his youth and strength much longer than most people. At 70, LaLane was able to tow 70 rowboats (some occupied) a mile in chilly, rough seas — an incredible two-and-a-half hour feat for a person of any age. He’s an anomaly of course. The real question for us mere mortals is whether we can achieve some degree of control over our aging and whether our bodies and cells have their own clock that might be adjustable.
In October 2013, UCLA professor Steve Horvath introduced the science community and the world to his ‘epigenetic clock,’ a natural mechanism by which all of our cells seem to track our chronological aging by changing the structure, but not the sequence of our DNA. Some believe this “clock” may play a significant role in the progression of some diseases while others hypothesize it’s merely a passive observer of time. In the short time since its discovery, much has been learned about how our cells monitor the passing of time and what environmental conditions may influence this molecular time keeper.

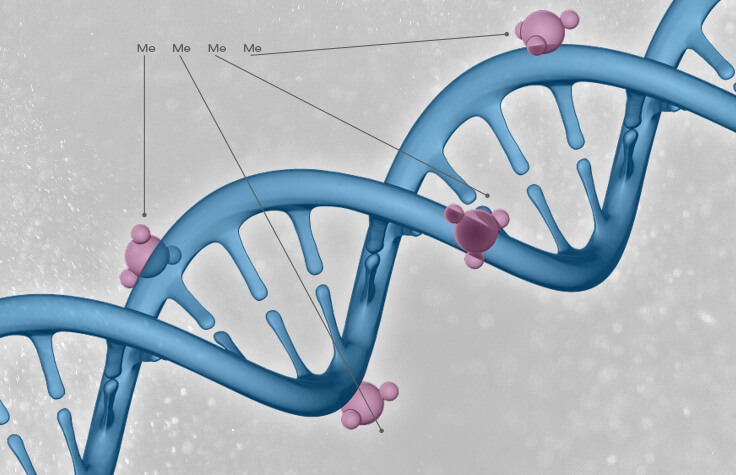
Epigenetics describes a variety of processes and influences that affect the way genes function and behave (i.e. their activity) without changing the physical sequence of the gene (which is what separates epigenetics from say a genetic mutation). Cells create these epigenetic marks by adding small molecules either to the DNA itself or to the proteins that are associated with the DNA (like histones).
To create his clock, Horvath used arguably the most well studied epigenetic process, methylation, in which a small molecule is directly added to the DNA. The affect is generally to decrease or cease the activity of a gene. He used genetic and methylation data collected from thousands of human samples, from in utero babies to 101 years olds, identifying 353 markers in the human genome. Using these markers, Horvath was able to create, a simple, predictive algorithm that allowed him to accurately determine a person’s chronological age by looking at the methylation status at each of the 353 markers.
The way the clock works is that at these sites in the genome, these specific 353 marks, can either have a methyl group or not (see picture below). Horvath’s algorithm uses takes into account pattern and found it correlated with chronological age, in other words by looking at these 353 sites, Horvath can very accurately guess your chronological age. At the time he said of his clock:
It’s surprising that one could develop a predictive tool that reliably keeps time across the human anatomy. My approach really compared apples and oranges, or in this case, very different parts of the body — including brain, heart, lungs, liver, kidney and cartilage.
But not all tissues correlated exactly to chronological age. Some tissues samples matched a pattern at these markers of a person much older. This was most notably seen with breast tissue, which was consistently 2-3 years older on the epigenetic clock than the rest of the tissue in a women’s body. Furthermore, the healthy tissue near the tumor was on average 12 years older than the rest of the tissue. Tissue from tumors on the whole was about 36 years older than the healthy tissue.This means the tissue’s pattern at Horvath’s markers matched that of a much older person. Many have suspected these discrepancies, once fully understood, could have important implications for the development of breast cancer.
The clock’s rate can also vary considerably based on age of the person and of a cell. Unsurprisingly, embryonic stem cells showed basically no epigenetic age, but after birth the epigenetic clock accelerates rapidly through adolescence and the teenage years. At about 20 years old, the clock slows down to a constant rate. At the end of our life span the clock tends to speed up and is a good indicator of mortality, as a five-year difference between your epigenetic clock and chronological age increases mortality risk by 20 percent. Even when correcting for several factors like age education, social class, hypertension, diabetes and cardiovascular disease, the mortality risk is 15 percent for a five year difference.
The epigenetic clock has also moved beyond Horvath’s initial proposal. Other epigenetic processes like chromatin (a packaging complex of DNA and associated proteins) modification have also been found to be associated with chronological age. Trey Ideker from the University of California San Diego, developed a similar clock to Horvath’s. The main difference is that Ideker’s clock uses far fewer methylation sites, just 71, which may make it more adaptable (i.e. cheaper and easier to use) to clinical applications. Ideker’s clock has found that kidney, breast, lung and skin cancers are on average 40 percent older than the patients from which they were removed. Brain cancers in particular were found to be decades older than the rest of the tissue in a body.
Time keeper or potential health care boon
Because of the apparent increased rate associated with cancer, the epigenetic clock could help physicians target which pre-cancerous tissue needs to be treated or help determine more accurately life expectancy for a person with terminal cancer. Both Horvath and Ideker see their clocks most useful application as a general monitor of health in old age. If a person’s epigenetic clock all of a sudden rapidly accelerates, it could be a potential sign that something is wrong and medical intervention or end of life care is necessary.
Cancer isn’t the only condition the clock appears to be influenced by as epigenetic age is accelerated by HIV status. Patients with detectable viral loads are older, epigenetically, than those who are healthy and those who are HIV positive but have their condition under control. The clock could become an important way to help manage and monitor the care of patients with HIV.
The clock’s utility may not be solely focused on human health. Forensic scientists believe the clock could be a potential aid in determining the age of an unidentifiable victim or of an unknown assailant. Ideker and collaborators are trying to perfect his epigenetic clock so it can work on small samples of blood for this purpose.
Scientists have known that epigenetic modifications like methylation are influenced by environmental factors, like nutrition. But it’s still unclear how this influences the methylation sites that are used in either of the epigenetic clocks. Horvath, as well as other labs, have found that obesity can accelerate the epigenetic clock of liver cells, which suggests that diet can influence the clock. Many lifestyle inputs like diet and exercise have been linked to alterations to chromatin, a packaging of DNA and proteins that can dictate how easy or hard it is for a gene to becoeme activated. But precisely how all these influences affect the epigenetic clock and aging, and whether or not these alterations are reversible, is still poorly understood.
The main question: is the clock an active or passive player in a cell. If the clock is active than there might be some therapeutic value as drugs that can alter the epigenome at the age marker could be used in cancer treatment. But if these markers are added as a result of cancer development and not as a part of it then there resetting the clock won’t hold any value here. The same is true of aging.
However, thus far more evidence has turned up to suggest the clock is merely a time keeper. Horvath’s initial belief was the clock may be malfunctioning in rare diseases where a person fails to age properly. In these instances, subjects appear to be arrested in development at a very young age, despite their chronological age being much older. Unfortunately in these cases, the epigenetic clock showed no discrepancy between chronological and epigenetic ages, in other words a faulty epigenetic clock was not the cause of the aging disorder. This seems to reinforce the clock’s role as a mere time keeper.
Yet even if this is the case, a time keeping role would be an invaluable tool for many fields. Further study and development of the epigenetic clock could help improve many aspects of health care and potentially other fields like criminal justice.
Nicholas Staropoli is the associate director of GLP and director of the Epigenetics Literacy Project. He has an M.A. in biology from DePaul University and a B.S. in biomedical sciences from Marist College. Follow him on twitter @NickfrmBoston.