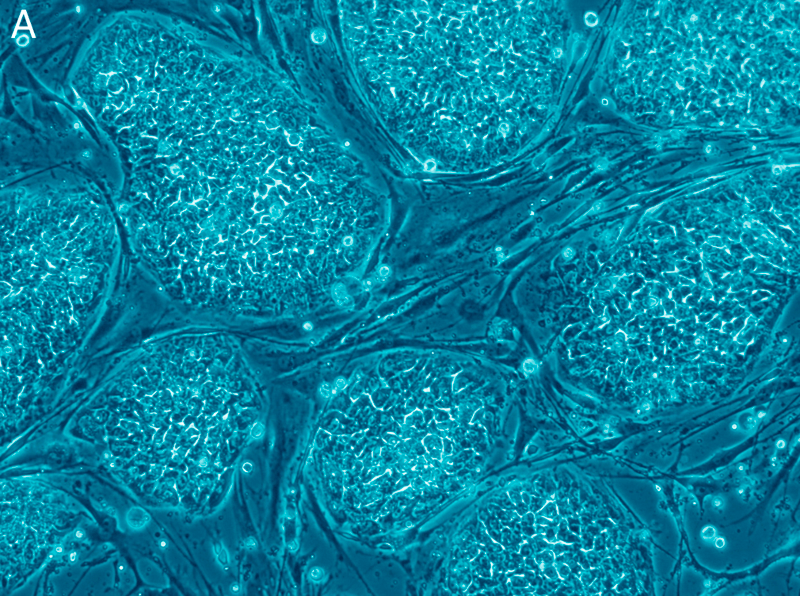
The secret behind the much-vaunted pluripotency of stem cells might lie in ancient viral DNA that was folded into the human genome long ago, according to a study published over the weekend in Nature Structural & Molecular Biology.
Stem cells — specifically, embryonic stem cells — are such a hot topic in medicine because of their ability to develop into any type of cell in the body. Think of it this way: your body had to grow all of its limbs, tissues, and bones at least once. Why, then, can we not harness this ability to grow them again to fight disease or repair damage? If we can find ways to “trick” our cells back into a pluripotent state, we might just be able to.
Despite the intense research surrounding stem cells, how their pluripotency works has remained unclear. Now it seems like our stem cells depend, at least in part, on genetic material “borrowed” from viruses long ago.
Some viruses, called retroviruses, can slip their own genes into DNA of their hosts. The most famous retrovirus is HIV. Most of the time, retroviruses only infect body cells, not sex cells used in reproduction; the retroviral genetic material exist only in the infected individual.
Every now and then, though, retroviruses do manage to slip into sperm or egg, and their genetic material gets passed down across human generations. Charles Q. Choi at National Geographic writes:
Past research suggests that at least 8 percent of the human genome is composed of these so-called endogenous retroviruses-leftovers from retroviral infections our ancestors had millions of years ago.
Scientists long thought that endogenous retroviruses were junk DNA that didn’t do anything within the human genome, said study co-author Huck-Hui Ng, a molecular biologist at the Genome Institute of Singapore.
Imagine their surprise when recent studies showed that one class of endogenous retroviruses, called HERV-H, was abundant in embryonic stem cells and seemed to accurately indicate pluripotency.
The latest study tested the potential role of HERV-H in pluripotency by taking human embryonic stem cells and treating them with bits of RNA that suppressed the activity of HERV-H. The treated cells lost their pluripotency; in essence, they stopped being stem cells.
So it seems clear that something important is happening with this viral DNA and our embryonic stem cells … but there’s an evolutionary angle to this mystery as well. HERV-H is unique to humans and primates, a relatively recent addition to our genomic menagerie of endogenous retroviruses. Other forms of life are creating pluripotent stem cells all the time without this genetic material.
Jason Mick at DailyTech goes into the potential evolutionary whys and wherefores of HERV-H, writing:
While I found this study interesting, I think it’s worth noting that the authors have yet to fully explain why mankind relies HERV-H for pluripotentcy. One possibility I would propose is that perhaps the gene was useful in dealing with the increasing complexity in primate tissues (primates have considerably more complex organ systems that a rodent, for example).
As far as explaining, in evolutionary terms, HERV-H’s transformation from viral invader to useful — arguably essential — component of the primate genome, this study is lacking.
The implication for endogenous retroviruses, though, is scintillating. Once a retrovirus merges with our genetic material by invading a sex cell, nothing stops evolution from acting on this new source of variation. There’s a wonderful irony in taking an invader that co-opts our cells to do its reproduction and co-opting it to somehow improve our own reproduction.
Hopefully, this study piques the interest of other genome researchers to look at more of our viral genomic hangers-on and see if they also seem to serve important, unexpected functions. And if we can figure out how and why HERV-H is essential to pluripotency, this might be one more step toward the goals of regenerative medicine.
Kenrick Vezina is Gene-ius Editor for the Genetic Literacy Project and a freelance science writer, educator, and amateur naturalist based in the Greater Boston area.
Sources:
- “Ancient Virus DNA Gives Stem Cells the Power to Transform,” Charles Q. Choi | National Geographic
- “Primate Stem Cell Creation Appears Driven by Genes From Ancient Virus,” Jason Mick | DailyTech
- “The retrovirus HERVH is a long noncoding RNA required for human embryonic stem cell identity,” Nature Structural & Molecular Biology