What if we could use the same ‘genetic recipe’ as lizards to regrow lost limbs the way a skink or gecko might regrow its tail after a dramatic bit of self-amputation meant to distract a predator? If the idea seems familiar, it might be because of the 2012 Spider-Man reboot; this is the exact conceit that leads the fllm’s antagonist to accidentally transform himself into a gigantic anthropomorphic lizard. (This has actually been The Lizard’s origin story since his comic debut in 1963.) Now a study out of Arizona State University suggests that using lizards as a genetic model to unlock our body’s own regenerative potential — sans supervillainy — might be possible.
 Lizards are, as Genetic Engineering & Biotechnology News has pointed out, far from the only creatures capable of regrowing limbs. They’re hardly even the most impressive when it comes to self-regeneration. Zebrafish can regrow lost fins; newts and other amphibians can regrow more than a simple tail but actually entire limbs. Some starfish can even regrow an entire body from a single limb. So if we’re in pursuit of regenerative abilities, why lizards and not any of these others?
Lizards are, as Genetic Engineering & Biotechnology News has pointed out, far from the only creatures capable of regrowing limbs. They’re hardly even the most impressive when it comes to self-regeneration. Zebrafish can regrow lost fins; newts and other amphibians can regrow more than a simple tail but actually entire limbs. Some starfish can even regrow an entire body from a single limb. So if we’re in pursuit of regenerative abilities, why lizards and not any of these others?
Put simply, we’re more like lizards than any of the other options. Specifically, the genetic machinery that allows for the spectacular regeneration of starfish (or newts, or zebrafish) is not present in our genomes. But “lizards basically share the same toolbox of genes as humans,” lead author Kenro Kusumi, professor in ASU’s School of Life Sciences said in a press release. “Lizards are the most closely-related animals to humans that can regenerate entire appendages.”
Kusumi and his team studied the 25-day process of tail regrowth in the green anole, a familiar little green lizard in pet-stores that is widespread across the Southeastern U.S. and Caribbean that will readily shed (and later regrow) its tail in self-defense. They tracked the sixty-day process of tail regrowth at a genetic and a cellular level. “We discovered that they turn on at least 326 genes in specific regions of the regenerating tail, including genes involved in embryonic development, response to hormonal signals and wound healing,” Kusumi said. Roughly 300 of these genes may also exist in humans in analogous form.
The researchers also produced an image showing the thin slices of tail at various stages of reproduction. Here’s the image, accompanied by a play-by-play from Wired’s Nick Stockton:
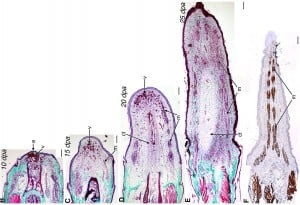
For the first 10 days, the lizard’s tail heals similarly to any other wound—new blood vessels form, and epithelial tissue (the soft, white skin under a scab) closes the gap over raw flesh. Then the tail starts to regrow, beginning with nerve tissue from the spinal cord (days 10-15), soft muscle, and tissue for transporting fluids. By day 20, the muscular tissue is starting to firm up around a cartilaginous tube[…] The far right of the image shows a 25-day-old tail. The lower-case ‘m’ indicates strong muscle fibers capable of quick motor control—the anole can flick its new tail.
A few caveats: lizards’ regenerative ability is not perfect. The new tail will not have bone, instead it will be supported by a rod of cartilage. It will also likely be a different shape color or different length  — it may even have odd patterns. (Here’s a yellow leopard gecko whose tail has grown back in the more traditional spotted coloration.) Furthermore, the tail is a relatively simple appendage for lizards that regrow it — it only really needs to be able to store fat and twitch. It’s not particularly analogous to the biomechanical complexity of a human arm and hand, with all its important little bones and fine musculature. It’s hard to imagine a functional human limb built around a simplified tube of cartilage.
— it may even have odd patterns. (Here’s a yellow leopard gecko whose tail has grown back in the more traditional spotted coloration.) Furthermore, the tail is a relatively simple appendage for lizards that regrow it — it only really needs to be able to store fat and twitch. It’s not particularly analogous to the biomechanical complexity of a human arm and hand, with all its important little bones and fine musculature. It’s hard to imagine a functional human limb built around a simplified tube of cartilage.
Upon closer examination, what makes this study interesting isn’t necessarily the “regrowing lost limbs” hook — though that is incredibly fascinating. It’s especially interesting in the context of regenerative medicine research, the brunt of which is focused on stem cells and recreating the same circumstances that allowed us to grow our limbs in the first place.
Well, anoles have already ‘figured out’ how to regrow an entire tail — albeit an imperfect one — even in adulthood, using a similar set of genes to the ones we have and without recapitulating their embryonic development. Perhaps we don’t need to build regenerative medicine from the ground up — our animal kin may have some blueprints we can use.
Kenrick Vezina is a freelance science writer, educator and naturalist in the Greater Boston area.