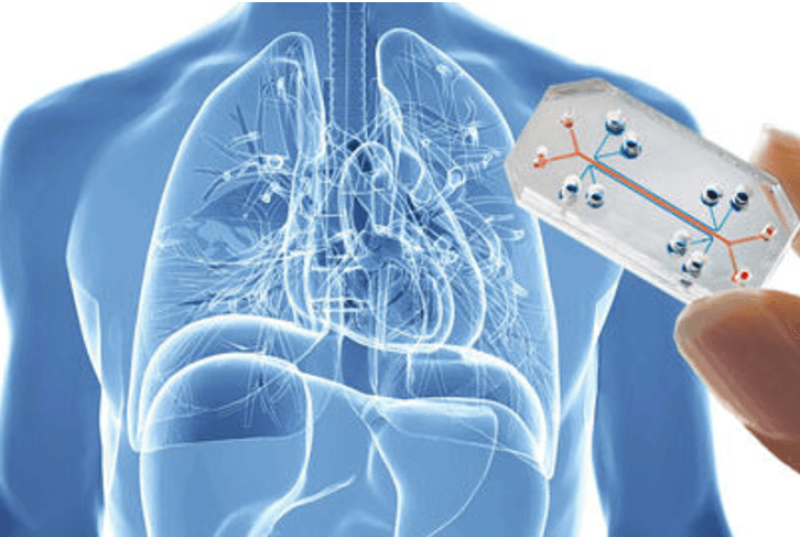
Disasters [in human drug trials] are rare. But also rare are clinical trials that go well: Ninety percent of drugs tested on humans do not work. These failures are an expensive but necessary part of the estimated $2.6 billion it costs to bring a medicine to market.
A lab in California is working on a more efficient and humane process, based on a quarter-size plastic slide. It’s a tiny replica of the human brain, rendered in clusters of about 300,000 cells surrounded by small copper wires. Based on that, Lawrence Livermore National Lab (LLNL) researchers have set out to build a comprehensive human body in miniature. Called the iChip, it could be the future of drug testing.
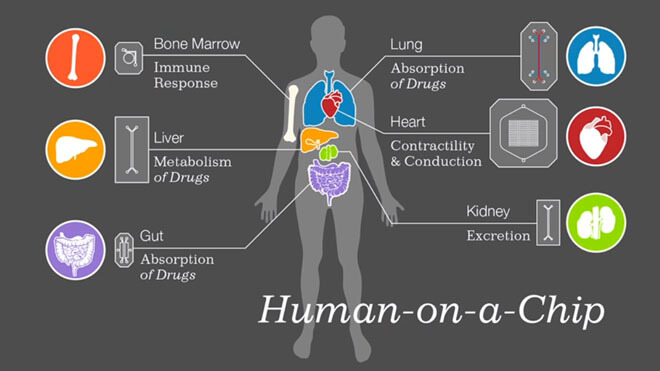
“Most people aren’t really thrilled with us injecting anything new into a human,” says Heather Enright, an iChip researcher. But testing drugs on animals instead provides limited information. For example, tumors develop differently in mice than they do in humans. With iChip, a dose of a new medication could be injected into the cells of a mini organ, and researchers could monitor the effects without harming live subjects.
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion, and analysis. Read full, original post: How to Conduct Human Drug Trials Without Needing Any Pesky Humans