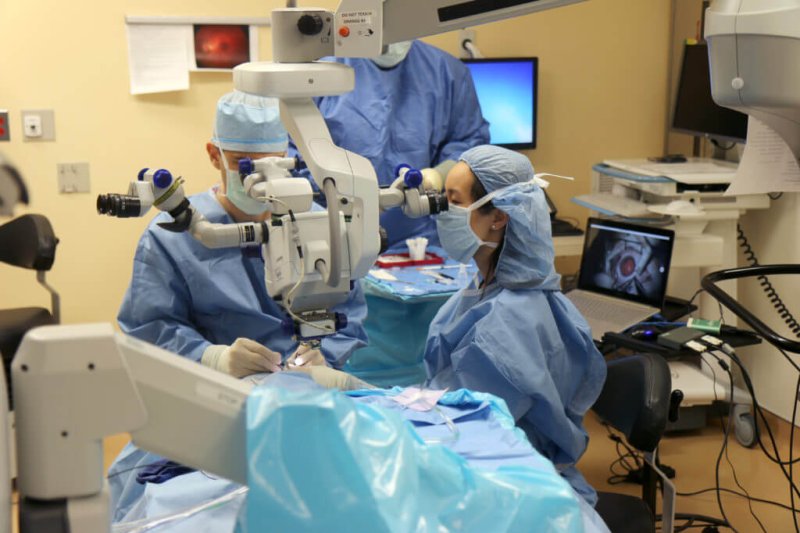
The trouble had started over a decade ago, when the Hogans noticed something wasn’t right with their son Jack…When they asked him to pick up his toys, he wouldn’t — not because he didn’t want to, it turned out, but because he didn’t have the peripheral vision with which to see them strewn across the floor.
…On Tuesday [March 20], he was to be the first person in the U.S. to receive a gene therapy for a rare inherited disease since the treatment had hit the market.
…
And they’d watched as the price was announced at $850,000 for both eyes — a record price so high that Spark Therapeutics worked out a way for insurers to pay in installments, as if the medication were a fancy car.
…
The treatment, developed after decades of research, is called Luxturna, and it is sometimes referred to as the first “true” gene therapy to be approved by the Food and Drug Administration.
…
Just then, the same surgery was just getting started on another patient in Los Angeles, and the next day it would happen again in Miami. For [ Dr. Jason Comander, Jack’s surgeon at Mass. Eye and EarComander] and the Hogans, there was still the other eye to do next week, and it was still up in the air as to whether the treatment would work at all. (Spark, to help win acceptance of the treatment’s high price, has set up a program to provide some kind of refund in case it doesn’t.)
Read full, original post: ‘That’s $425,000 right there’ — the anxious launch of a gene therapy with a record sticker price