The work is the first protein therapy for a genetic condition delivered via the amniotic fluid. But like familiar protein therapies that are injected or infused, such as insulin and clotting factors, the protein must reach the bloodstream. It does so, thanks to a clever commandeering of the molecules that enable a fetus to grab maternal antibodies through the placenta before birth, and in mother’s milk after.
 Timing is critical
Timing is critical
Like most, if not all, medical breakthroughs, prevention of XLHED symptoms is the culmination of years of experiments. But this story is unusual in that disappointing clinical trial results didn’t halt the work, but revealed that what was awry was the timing of the treatment – not the treatment itself.
During development, each anatomical structure has a “critical period” when it is sensitive to the actions of genes and environmental insults. That’s why millions of women who took the drug thalidomide in Europe during early pregnancy in the early 1960s gave birth to babies with stunted or absent limbs – the drug acted precisely when the arms and legs telescope out from buds.
In mice and dogs with XLHED, delivery of a replacement protein just after birth prevented symptoms. But the structures that give rise to teeth, sweat glands, and hair – from the outer layer of the embryo, the ectoderm – form earlier in human fetuses, in the middle of gestation.
Distinctive symptoms
Hypohidrotic ectodermal dysplasia affects at least 1 in 5,000 to 10,000 newborns, with the type transmitted on the X chromosome the most common form. Genetic testing can identify mutations and ultrasound scans reveal the telltale absence of tooth buds.
A boy with XLHED has scant, light, and dry hair, missing or pointy teeth, and dry eyes, skin, mouth, airways and mucous membranes due to abnormal development of exocrine glands. The inability to sweat can be dangerous and even deadly, particularly in a newborn coping with a fever or heat wave. The absence of teeth may disrupt the jaw so greatly that creating implants or fixing the teeth that do erupt is difficult if not impossible.
The gene that is mutant in XLHED encodes a protein, ectodysplasin A1 (EDA-A1), which binds a receptor, ectodysplasin receptor (EDAR), on the surfaces of cells destined to develop as hair, teeth and sweat glands. The binding triggers signaling pathways inside the cells that control the interaction of parts of the ectoderm with parts of the middle layer of the embryo, the mesoderm.
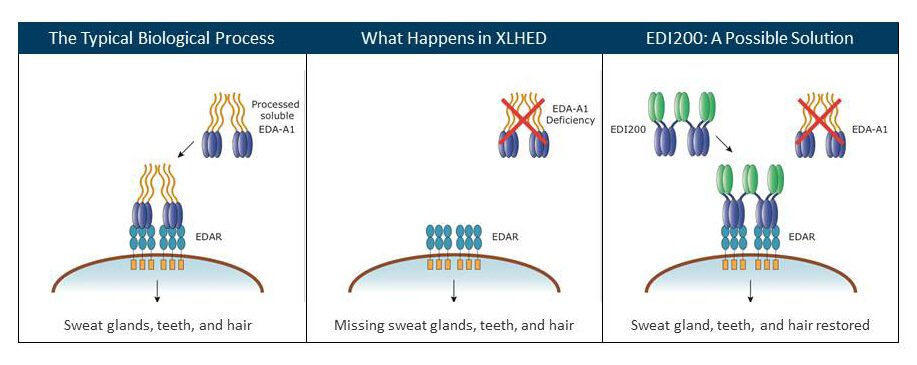
In XLHED, EDA-A1 can’t bind its receptor, blocking the sending of signals for hair, teeth and sweat glands to develop. The new treatment replaces the part of the protein that targets the receptor with a segment of antibody molecule immunoglobulin G (IgG). The part, called Fc, acts somewhat like a guidance system on a drone.
“The Fc is key to cross some barriers by hijacking the IgG transport system: IgGs are either transported through the placenta (to load the fetus with maternal antibodies before birth), or through the intestinal epithelium (to transport maternal antibodies present in milk to the circulation of the newborn). In both cases, the aim is to transfer maternal humoral immunity to the baby,” explains Dr. Schneider. In its engineered role, Fc attached to the guiding part of EDA-A1 replaces the faulty signal behind XLHED.
In work published in Nature Medicine in 2003, Olivier Gaide and Pascal Schneider, at the University of Lausanne in Switzerland, created a recombinant fusion protein consisting of EDA-A1 and Fc, growing it in cultured mouse cells. They introduced the fusion proteins into the circulations of pregnant “Tabby” mice that have the condition (the human and mouse EDA proteins are identical). The treatment prevented symptoms, and did so in dogs too, but given earlier in pregnancy. Dogs don’t sweat but they transfer measurable moisture through their paw pads.
First clinical trial
With mice and dogs responding, researchers hypothesized that giving the doctored protein to newborns with XLHED might correct the stymied developmental signal in time to jumpstart formation of tooth buds and sweat glands. Edimer Pharmaceuticals got as far as treating 10 infants in the US and Europe in a phase 2 clinical trial, from 2013 to 2015.
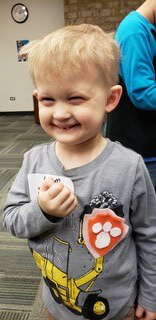
Three-year-old Liam Orchard Kudwa is one of the ten boys. Unlike trial participants who were at risk because of XLHED in their brothers, Liam’s mother, Beth Orchard, has the condition. She’s a blogger; read her story here.
Women are unlikely to have symptoms associated with recessive mutations on the X chromosome because they have a second X. But in all cells of female mammals, one X chromosome is shut off. If, by chance, in a heterozygote like Beth, many more cells have the normal X turned off, symptoms of the condition can arise. She didn’t know that her fine hair, scant sweat, and missing and cone-shaped teeth were part of a syndrome until her late twenties.
Beth began attending meetings of the National Foundation for Ectodermal Dysplasias (NFED) long before she and her husband Steve Kudwa had children, where they learned about the planned clinical trial. They enrolled Liam right after his diagnosis shortly after birth, and he received three doses of the protein, starting at two weeks of age, over a two-week period at St. Louis Children’s Hospital. Although the treatment was invasive, Beth recalls feeling guilty that her son was so healthy compared to the other babies in the NICU.
But the researchers already had an inkling from the animal studies that earlier intervention — before birth — might have been a better approach. And so Beth and Steve hoped that Liam would evade the symptoms, but didn’t expect it. “Two months, 4 months, 6 months, a year went by. The markers weren’t showing improvement in the treated children: sweat glands on the feet, number of tooth buds, jaw development, and the distinctive craniofacial features of XLHED,” Beth said.
“Trial to Cure”
The researchers stopped following the ten boys after a year, and the pharmaceutical company ran out of funding. But company researchers continued to help Dr. Schneider and his team work on providing the therapy in utero on a compassionate use basis, called “Trial to Cure,” for a few fetuses, at University Hospital Erlangen. This time around, the plan was to use the same fusion protein from the initial clinical trial, but delivered into the amniotic fluid, during the time (weeks 20 to 30) when the sweat glands form.

In early 2016, an obstetrician referred a 38-year-old woman pregnant with twin boys to Dr. Schneider. The mother was concerned that the fetuses had inherited XLHED, which her older son Joshua has. An ultrasound showing the absence of tooth buds in both twins confirmed her fear.
Four weeks later, a physician removed about 3 teaspoons of amniotic fluid from the woman’s belly and replaced it with an equal volume of the fusion protein. (That’s just 15 milliliters compared to the 500+ milliliter capacity at 26 weeks of gestational age.) The twins swallowed the groundbreaking treatment, and did so again at 31 weeks.
Through the fetal intestines the fusion protein reached their bloodstreams, where it needed to be to reboot development of the ectodermal derivatives. But the protein didn’t get into the mother’s circulation, demonstrating the safety of the treatment.
A few months later, another pregnant woman and her 19-week fetus joined the “Trial to Cure” experiment. Ultrasound showed he had no tooth buds at all. He received only one dose because that’s all that was left from the clinical trial, but he sprouted 9 tooth buds! He’s 14 months old now, but his sweating isn’t entirely normal.
By December 2016, twins Linus and Maarten seemed on their way towards the normal crying and drooling of teething. A post from NFED reported that the twins were sweating, salivating, and seeing well, with normal signs of 8 and 10 teeth set to erupt. And they weathered two very hot summers. Meanwhile, their brother couldn’t sweat and had only 3 teeth and one tooth bud.
 A one-time fix?
A one-time fix?
What will happen when it’s time for the tooth fairy to visit the three boys? Will adult teeth push upwards? Time will tell.
“When EDAR is stimulated, the signal is given and the structure can develop in the absence of further EDA signals. Thus, in principle, no more treatment is required. However, not all EDA-dependent structures develop at the same time, and repeated treatment might be necessary to stimulate formation of appendages that develop at earlier or later stages. For example, it might be necessary to repeat treatment if one wants to get a full set of secondary teeth, including wisdom teeth. Whether several treatments are required for this, and if yes when treatments should be administered, needs to be investigated further,” Dr. Schneider said.
And protein therapy isn’t gene therapy – Linus and Maarten and the third boy will pass the mutation that is part of their X chromosomes on to their children.
The ability to introduce healing proteins at critical times in development via amniotic fluid will extend to other selected conditions. “The same approach could be applicable, for example, for certain genetic orofacial clefts which are not that rare. Administration of the missing protein could then lead to cleft closure in utero without the need for postnatal surgery,” said Dr. Schneider.
In an editorial, Marja L. Mikkola, PhD, of the University of Helsinki calls the results “remarkable and encouraging.” Indeed they are.

Beth Orchard, who is very active with NFED, knew the good news ahead of this week’s publication, although she missed the update and the opportunity to meet Dr. Schneider at last summer’s conference because she was busy giving birth to Iona.
She’s thrilled. “It’s exciting to hear such positive results. We had no preconceived notions that the therapy would cure Liam. We see his few teeth come in, see him struggle with eating, and feel a little bad. But at the same time, he was part of something bigger. Without him and the other boys, Dr. Schneider and his team couldn’t have done the next part. Hopefully they’ll now do a bigger study and after that other things will come down the road. Science being what it is, the work will contribute to breakthroughs for other syndromes, not just XLHED.”
A loud shout-out for all of the fearless and tireless family advocates in the rare disease community!
Ricki Lewis is the GLP’s senior contributing writer focusing on gene therapy and gene editing. She has a PhD in genetics and is a genetic counselor, science writer and author of The Forever Fix: Gene Therapy and the Boy Who Saved It, the only popular book about gene therapy. BIO. Follow her at her website or Twitter @rickilewis.































