My background is in molecular biology – the study of DNA, genes and how an organism’s blueprints are decoded and assembled into life. The discipline requires an understanding of how molecular codes are deciphered and turned into functional biology. Anyone in this field is plagued with dreams of dancing molecules, interacting and performing the roles that turn DNA information into our food, the plants in our environment and our families.
Every day in the lab we move genes around. It’s easy. Not meant to generate new products for consumers, moving DNA is used as a research tool that lets us understand how specific genes work. A classic example is the NPR1 gene from the model plant Arabidopsis; it’s a defense gene that confers enhanced tolerance to disease when you drop it into almost any plant’s genome. Manipulating genetic information – in plants, microbes and some animals – is commonplace.
On that half-cut lawn it occurred to me – instead of inserting DNA information we understand, what if we introduced a scrambled mess of random DNA code into a plant or bacterium? Could we identify random bits of genetic information that could give rise to small proteins (called peptides) that change an organism’s physiology or development?
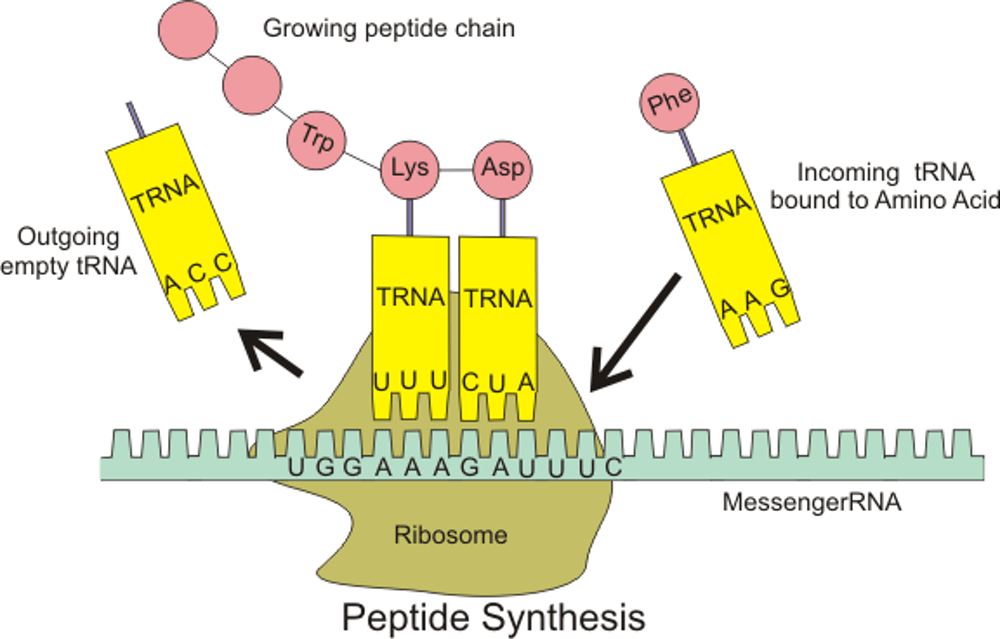
Normally DNA encodes instructions that coordinate the order of the amino acid building blocks in a protein. Each amino acid has specific chemical characteristics. Strung together in a peptide chain, they fold into a protein that provides cellular structure or function, based on the complementary chemistries of its amino acid components.
My hypothesis was that a short, scrambled DNA message could give rise to a novel string of amino acids. This would be a small cluster of discrete chemistry that likely never existed before on the planet. The vast majority of the time it would be meaningless and just become cellular rubbish. But maybe on rare occasion it could do something new and desirable.
To test the hypothesis, our research team used randomized templates to synthesize trillions of random DNA fragments using simple DNA amplification techniques. Each was flanked by the genetic instructions to start and stop production of a peptide inside the plant.
Then we used standard genetic engineering techniques to insert a novel DNA sequence into thousands of individual Arabidopsis thaliana plants – and sat back to watch what would happen when the plants turned the random genetic information into little random peptides. We were hoping for cases where specific protein structures might find a connection with biological chemistry and we’d see the result in the plants themselves.
As the plants grew, we were blown away by what we observed.

Some plants were flowering early. Others were small and stunted. Others grew larger leaves. Some were loaded with healthy purple pigments. Still others grew up to a point…then died.
We then retrieved the particular random DNA sequence we’d added to each, a simple feat for a molecular biologist, and inserted the same sequence into new plants. Most of the time the random information affected the new generation of plants in exactly the same way, demonstrating that something was indeed happening related to the added, garbled information. We published our results in the journal Plant Physiology.
What is this random information doing inside the cell? The small random molecules generated from the inserted DNA instructions could affect a specific process, just by chance. They could bind a needed nutrient. They might inhibit a key enzyme. They could turn on flowering or protect a plant from freezing. Nobody really knows exactly how until the plants are examined in detail one by one. These new proteins may also be good models to design new useful molecules with similar chemical properties, but that are more durable in the cell. Our goal is to produce a compound that may be applied to crops to change the way plants grow and behave, or perhaps stop the growth of invasive or weedy plants.
The process is like throwing monkey wrenches into a complicated machine. Most of the time they clank around and affect nothing; but once in a long while a wrench catches in some critical gears and brings the machine to a halt. Other times the wrench might short-circuit a wasteful process, allowing the machine to run more efficiently. These peptides are molecular monkey wrenches.

Some of these peptides must interfere with an important biological process because they kill the plant. These findings bring to light new vulnerabilities in plants that researchers could exploit to develop environmentally friendly and nontoxic herbicides. Agriculture currently relies on a few relatively old chemistries, cultivation (using fossil fuels) or human labor to control the weeds that compete with food plants for resources. Good weed control means that valuable fertilizers, water and sunlight go only to the desired plants, rather than weeds. So new herbicide chemistries would be extremely valuable as farmers work to produce food for growing populations.
But why stop at plants? We are using the same approach to discover the next generation of antibiotics. The goal is to identify random information that affects a single species of problematic bacterium. For instance, we could potentially target S. aureus, the antibiotic-resistant bacteria that causes MRSA. We are hunting for new molecules that could destroy MRSA-related bacteria while leaving the rest of the microbiome unaffected. These experiments are underway in our lab.
Randomness may pinpoint undiscovered vulnerabilities or opportunities in plants, bacteria and other organisms. There even may be applications in solving human disease. The future is exciting as we mine the vast collections of new molecules and study how they integrate with biology to produce important desired outcomes.
Several of the molecules we’ve already identified slow plant growth. Future products from this technology might even be applied to make lawns grow more slowly. While others may find this advance helpful, I’ll have to skip using it. Cutting the grass gets my good ideas flowing.
A version of this article was originally published at The Conversation as “Can random bits of DNA lead to safe new antibiotics and herbicides?” and ran on the GLP on Sept. 27, 2017.
Kevin Folta is a plant physiology and molecular biology researcher. He currently works with farmers in order to understand agricultural technology and food security. Follow him on Twitter @kevinfolta































