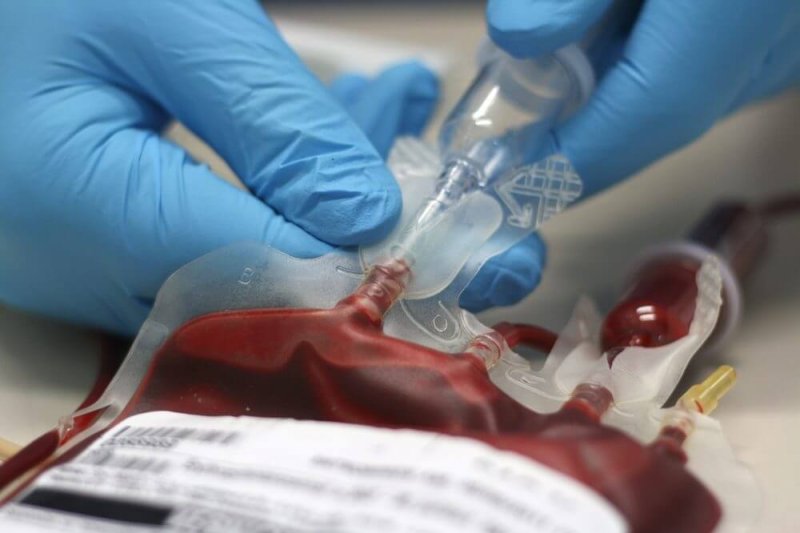
Biomarin Pharmaceutical Inc. has submitted a biologics license application to FDA and documentation of clinical trial results to the European Medicines Agency, with reviews slated to begin early this year at both organizations.
An article in the January 2 New England Journal of Medicine from a UK research team presents the findings of a phase 3 analysis of continuing success of a phase 1/2 trial (instead of a new phase 3 trial). The hemophilia gene therapy – called valoctocogene roxaparvovec for now – can mean a one-time infusion that replaces the more than 100-150 infusions of clotting factor a patient takes each year, and can also alleviate the painful joint bleeding that is the hallmark of the disease.
The different clotting disorders result from mutations in different genes in the pathway that knits a clot from protein fibrils. Hemophilia A is a deficiency of clotting factor VIII, and is also called classic hemophilia. It accounts for 80 percent of people with the disease. The clotting disorder that threaded through the royal families of Europe was hemophilia B, which is a deficiency of factor IX.
Both hemophilias are transmitted by genes on the X chromosome, and therefore affect only males. One in 10,000 males has hemophilia A, and it arises as a new mutation (rather than being inherited), in about a third of cases.
Ryan White’s story

The world focused on hemophilia A with the sad case of Ryan White. Born in 1971, Ryan was diagnosed at 3 days of age when his circumcision wound wouldn’t stop bleeding.
Like many people with hemophilia A at the time, Ryan received weekly factor VIII pooled from donors. That would prove tragic, as President Reagan was late to the game of testing the blood supply for viruses. He refused to even utter the word “AIDS” until actor Rock Hudson died of it in 1985.
That was too late for Ryan White.
HIV as the cause of the mysterious new epidemic was identified in 1983, although for a time it was known by different names.
In 1984, Ryan had a lung biopsy to diagnose severe pneumonia that revealed that he was HIV positive. Nearly 90% of people with hemophilia who received clotting factors from pooled donor blood between 1979 and 1984 contracted HIV and/or hepatitis C.
Ryan survived longer than predicted, until the end of 1990. In the intervening years he catalyzed AIDS activism when he was denied admittance to school, and he and his family fought the discrimination and ignorance.
Hemophilia A gene therapy has been twenty years in the making.
Don Miller’s story
By the end of the decade that began with Ryan White’s death, the first gene therapy for hemophilia A was being tested in a clinical trial in Pittsburgh. I was fortunate to interview the first patient soon after he received the gene therapy.
Like Ryan White, Don Miller had nearly bled to death when he was circumcised. He recalled other frightening incidents.
“I fell at my grandmother’s house and had a one-inch-long cut on the back of my leg. It took five weeks to stop bleeding. It leaked slowly, so I didn’t need whole blood replacement. But if I moved a little the wrong way, it would open and bleed again.”
Miller’s treatments paralleled the history of countering hemophilia, from whole blood infusions, to plasma replacement, to cryoprecipitate (a frozen plasma product containing clotting factors). Then he injected pooled factor VIII three times a month. But somehow he never contracted HIV, and that’s what got him into the gene therapy clinical trial.
“I lucked out,” Miller told me. Besides his good fortune at dodging the HIV bullet, he was in the right place at the right time. He’d been a librarian at the University of Pittsburgh, where the clinical trial was to take place.
On June 1, 1999, Don Miller received the first of three injections of retroviruses engineered to carry factor VIII. Chiron Corp., one of the original wave of big biotech companies that was absorbed into Novartis in 2006, designed and manufactured the vector.
The goal of this first round of hemophilia A gene therapy wasn’t to cure the disease, but to boost factor VIII levels a scant 2 to 7 percent, which was expected to dampen bleeds.
Don Miller hadn’t had any side effects when I spoke with him, but I don’t know how he fared. At the time, he spoke freely to the media, but I contacted his physician and she couldn’t provide an update due to HIPAA regulations.
But that first hemophilia A gene therapy was safe. Some patients had transient increases in factor VIII, and for 5 of the 13, bleeds became less frequent.
Retooled for a more lasting effect
When the gene therapy field emerged from several setbacks that began with the death of Jesse Gelsinger in 1999, the hemophilia strategy changed to a safer and more efficient vector. The clinical trial leading to the current FDA submission began with 15 patients who received the factor VIII gene delivered in adeno-associated virus serotype 5, starting in June 2015. A year later, 13 of the men treated with a single infusion had normal or near-normal levels of factor VIII.
 The “AAV5” vector coaxes greater expression of the gene than does the retrovirus used in the earlier trial. Plus, an added bit of control DNA (a promoter) directs the vector to the types of cells that normally make the factor in the liver and to white blood cells.
The “AAV5” vector coaxes greater expression of the gene than does the retrovirus used in the earlier trial. Plus, an added bit of control DNA (a promoter) directs the vector to the types of cells that normally make the factor in the liver and to white blood cells.
AAV5’s capacity is only about 4,700 DNA bases, so the big factor VIII gene is trimmed a bit (as it was for the earlier trial too). Still, it had to be delivered in two viral shipments, with some assembly required, like sending a cell phone and its charger in separate Amazon prime boxes.
Unlike other vectors (lentivirus and retroviruses), AAV remains separate from the cell’s chromosomes, forming a DNA circle called an episome.
Two of the 15 patients received doses too low (6 or 20 trillion viruses per kilogram of body weight) to have an effect. But six men receiving an intermediate dose of 40 trillion viruses had no bleeding events; the annual number of needed factor VIII infusions for the three-year study period fell from 155 to 0.5, and only one man had a bleed in a large joint.
The high-dose (60 trillion viruses) men did the best: none required factor VIII infusions, bleeding events, or large joint bleeds.
Analysis of factor VIII levels and various biomarkers of the gene indicated that of the 13 men who responded, one is considered to no longer have hemophilia, eleven have mild disease, and one has moderate disease.
It all adds up to what the researchers call a “sustained, clinically relevant benefit.” Said lead author John Pasi, from the Royal London Haemophilia Centre, Barts Health NHS Trust:
Our 2017 paper showed that gene therapy could significantly boost factor VIII levels in men with hemophilia A. Our new data are critical in helping the scientific and medical communities understand this pioneering technology. This latest study confirms both safety and long-term beneficial impact. A long-term treatment that effectively ends the life-long regular injections can transform care and massively improve the quality of life of hundreds of thousands of people born with this challenging genetic condition.
Once an advisory committee to FDA agrees, valoctocogene roxaparvovec will debut – with a catchier brand name. It may cost in the $400,000 to $1 million range (or higher) of other gene therapies in the US and Europe, but considering that current therapies for hemophilia A are about $270,000 annually without complications and can exceed $1 million if there are, a one-and-done gene therapy for hemophilia A sounds like a good deal.
Ricki Lewis is the GLP’s senior contributing writer focusing on gene therapy and gene editing. She has a PhD in genetics and is a genetic counselor, science writer and author of The Forever Fix: Gene Therapy and the Boy Who Saved It, the only popular book about gene therapy. BIO. Follow her at her website or Twitter @rickilewis