The Council negotiating mandate includes changes to the Commission’s proposal including:
- opt-out from cultivation: member states can decide to prohibit the cultivation of category 2 NGT plants on their territory
- optional coexistence measures: member states can take measures to avoid the unintended presence of category 2 NGT plants in other products and will need to take measures to prevent cross-border contamination
- the Council’s position also clarifies that, in order to avoid the unintended presence of category 1 NGT plants in organic farming on their territories, member states can adopt measures, in particular in areas with specific geographical conditions, such as certain Mediterranean island countries and insular regions
Category 1 new genomic techniques plants and patenting
- Under the Council’s mandate, when applying to register a category 1 NGT plant or product, companies or breeders must submit information on all existing or pending patents. The patenting information must be included in a publicly available database set up by the Commission which lists all NGT plants that have obtained a category 1 status. The database aims to ensure transparency regarding NGT 1 plants. If necessary, the information about patents included in this database must be updated.
- Furthermore, on a voluntary basis, companies or breeders can also inform of the patent holder’s intention to licence the use of a patented NGT 1 plant or product under equitable conditions.
- The Council’s mandate provides for the creation of an expert group on the effect of patents on NGT plants, with experts from all member states and the European Patent Office.
- According to the Council’s mandate, one year after the entry into force of the regulation, the Commission will be required to publish a study on the impact of patenting on innovation, on the availability of seeds to farmers, and on the competitiveness of the EU plant breeding sector. The study will also have a special focus on how breeders can have.
Labelling
- Category 2 NGT plants must contain a label indicating them as such, in line with the Commission proposal.
- The Council proposes that, in case information on modified traits appears on the label, it must cover all the relevant traits(e.g. if a plant is both gluten-free and drought-tolerant owing to genomic changes, either both of those features or neither of them should be mentioned on the label). The Council proposes this to ensure that consumers have access to accurate and comprehensive information.
Traits: The Council negotiating mandate states that tolerance to herbicides cannot be one of the traits for category 1 NGT plants. The Council proposes this change in order to ensure that such plants remain subject to the authorisation, traceability and monitoring requirements for category 2 NGT plants.
The European Parliament adopted its negotiating position on NGTs in February 2024, before the June 2024 European Elections.
MEPs want to keep mandatory labelling of products from both NGT 1 and NGT 2 plants. MEPs also agree that all NGT plants should remain prohibited in organic production as their compatibility requires further consideration and want the Commission to report on how consumers and producers’ perception of the new techniques is evolving, seven years after its entry into force.
NGT 1 plants: For NGT 1 plants, MEPs want to amend the size and number of modifications needed for a NGT plant to be considered equivalent to conventional plants. To ensure transparency, MEPs agree to set up a public online list of all NGT 1 plants.
NGT 2 plants: For NGT 2 plants, MEPs agree to maintain most of the requirements of the GMO legislation, which is among the strictest in the world, including the authorisation procedure. To incentivise their uptake, MEPs agree to accelerate the risk assessment procedure for NGT 2 plants expected to contribute to a more sustainable agri-food system, but underline that the so-called precautionary principle must be respected.
Ban on all patents filed for NGT plants: MEPs want a full ban on patents for all NGT plants, plant material, parts thereof, genetic information and process features they contain, to avoid legal uncertainties, increased costs and new dependencies for farmers and breeders. They also request a report by June 2025 on the impact of patents on breeders’ and farmers’ access to varied plant reproductive material as well as a legislative proposal to update EU rules on intellectual property rights accordingly.
Why should GE and GMO be regulated in different ways?
GE is done by a group of technologies that make the changes within the organism’s own DNA by moving, adding or deleting precise pieces of genetic material. Scientists argue that such changes are what happens in conventional breeding and can also come from induced mutations, as seen in the malting barley variety Golden Promise. In contrast to this, GM technologies involve inserting new DNA into an organism’s genome to introduce desired characteristics into plants or animals.
Background:
On 5 July 2023, the European Commission adopted a proposal for a new Regulation on plants produced by certain new genomic techniques (NGTs) which include advanced methods of gene editing, such as CRISPR-Cas9. These techniques hold promise for advancing agriculture, improving food security, and addressing environmental challenges. The EU’s proposal aims to update existing biotechnology regulations to provide a more streamlined framework for their application, while ensuring safety and transparency.
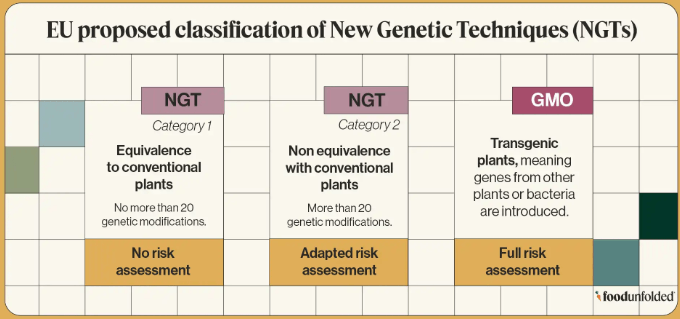
The proposal creates two distinct pathways for NGT plants to be placed on the market:
- category 1 NGT plants: could occur naturally or through conventional breeding methods; they would be exempted from the rules currently set out in the GMO legislation and would not be labelled; however, seeds produced through those techniques would have to be labelled
- category 2 NGT plants: all other NGT plants; rules under GMO legislation would apply (including a risk assessment and authorisation before they are placed on the market); they would be labelled as such
The proposal is accompanied by an impact assessment, supported by an external study, JRC case studies on several applications of NGTs and the scientific work of EFSA in the area of new genomic techniques.
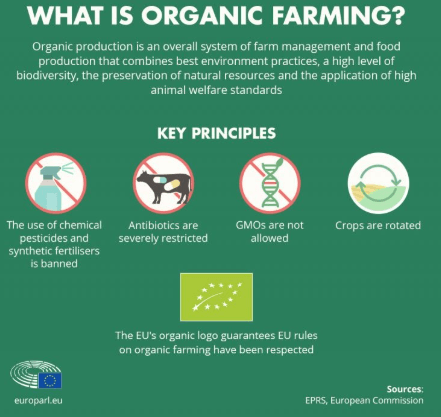
Organic: NGT plants will be prohibited in organic production. For NGT plants subject to authorisation, the legislative proposal maintains the traceability and labelling requirements of the GMO legislation. Today, GMOs are banned in organic production by the EU Organic Production Regulation. In addition, the proposal makes the adoption of coexistence measures at national level mandatory. Member States must adopt measures so that different types of cultivation can exist side by side, e.g., distances between the fields.
To exclude NGT plants from organic production, even those that have been verified to be comparable to conventional plants, organic and GM-free farmers can consult a public register of all NGT products and seed labelling in common catalogues of varieties.
What does this mean for the UK farmers?
Under the EU-UK TCA, Northern Ireland continues to follow EU regulations on plant health and safety, including the use of NGTs. The BAB team continues to work alongside UFU to ensure the implications for farmers in Northern Ireland are recognised withing this dossier.
Next steps:
The agreement on the Council’s negotiating mandate allows its presidency to start trilogue negotiations with the European Parliament on the final text of the regulation. We expect challenging discussions as MEPs will push back on patents, as well as labeling and safety assessments. The talks will be led on Parliament’s side by Swedish MEP Jessica Polfjärd from the center-right European People’s Party (EPP), a group broadly supportive of biotech innovation. The final outcome will need to be formally adopted by the Council and the Parliament before the regulation can enter into force.
Jenny Brunton is Senior European Policy Advisor at the British Agriculture Bureau. Follow Jenny on X @jennybrunton94
A version of this article was originally posted at British Agriculture Bureau. Find British Agriculture Bureau on X @UKFarmingUnions

































