The scientific community has been abuzz since the retraction of a controversial paper in Food and Chemical Toxicology by French scientist Gilles-Éric Séralini that allegedly showed a link between genetically modified corn and cancer, with allegations as to why it was retracted. Editor-in-Chief A. Wallace Hayes has now written a letter describing his thought process behind the retraction and addressing allegations by anti-GMO activists that his action was inappropriate.
Contrary to what has been suggested by some, the appointment of Professor Richard Goodman, University of Nebraska, as an Associate Editor was not influenced by Monsanto or any other party…It is understandable that Dr. Goodman’s involvement, however small, might be cause for concern for some. However, the decision to retract the paper was mine alone, made by me exclusively and not by a vote of the editorial board.
Hayes also responds to allegations that his decision to pull the paper violated guidelines for retraction under the Committee on Publication Ethics.
According to the COPE guidelines, “Journal editors should consider retracting a publication if… they have clear evidence that the findings are unreliable, either as a result of misconduct (e.g. data fabrication) or honest error (e.g. miscalculation or experimental error).”(COPE, 2009)….The review of the data made it clear that there was no misconduct. However, to be very clear, it is the entire paper, with the claim that there is a definitive link between GMO and cancer that is being retracted. In our analysis, his conclusions cannot be claimed from the data presented in this article.
He also addressed accusations by other activists that an unrelated safety study on the same corn by Monsanto, which found no safety concerns, was given special treatment and should be retracted as well. Hayes says he reviewed this paper in detail and found that it met COPE guidelines.
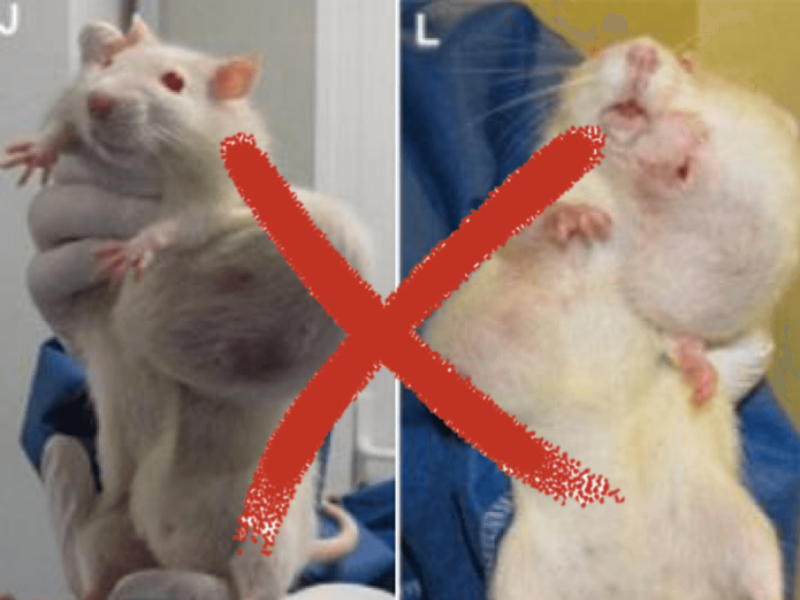
The Hammond et al. article was a 13 week feeding study performed in rats feed grain from Roundup Ready corn which is tolerant to the herbicide glyphosate (Hammond et al., 2004). The authors reported the following: “Purina TestDiets formulated Roundup Ready corn grain into rodent diets at levels of 11 and 33% (w/w). The responses of rats fed diets containing Roundup Ready corn grain were compared to that of rats fed diets containing non-transgenic grain (controls)…
In accordance with OECD Guideline No. 408 (OECD, 2009a), the Hammond et al. study was limited to 90 days following and used 20 rats/sex/group, and was conducted in general compliance with OECD Good Laboratory Practice (GLP) guidelines, as previously stated. The Séralini et al. study ran for two (2) years with only 10 rats/sex/group…While the number of animals used may have been sufficient to reach conclusions regarding oral toxicity, it proved insufficient for conclusions related to the carcinogenicity of the test substances.
Claire Robinson, editor of GM Watch and GMO Séralini, quickly responded to Wallace’s letter with another attack, making four key points:
[D]id Hayes actually read the Seralini paper before retracting it? Because if he did, how come he doesn’t know what the paper says? Hayes not having read the paper is, of course, the charitable interpretation of his actions, because if he is familiar with the paper’s contents it might be hard not to conclude that he has deliberately misrepresented them.Séralini‘s methodology was more rigorous than that of Monsanto’s, while generating just as much data per group.”
There is nothing in the COPE guidelines that justifies or requires the retraction of the whole paper.
The fact that … former Monsanto employee [Richard Goodman] was involved in discussion of the (re)review of the Séralini paper from the start, and up until Séralini directly requested that this stop, means that he could not only have influenced the discussion but even more crucially the selection of the review panel members.
Read the full, original story: Food and Chemical Toxicology Editor-in-Chief, A. Wallace Hayes, Publishes Response to Letters to the Editors
Additional Resources:
- French scientist Séralini threatens suit after discredited anti-GMO paper retracted, Genetic Literacy Project
- Food and Chemical Toxicology’s bungled handling of Séralini publication fiasco permanently damaged biotech movement, COSMOS
- Analysis of Séralini data shows why the botched GMO rat study was retracted, Pharyngula