Actor Michael J. Fox has had Parkinson’s disease for many years. It hasn’t kept him from continuing his professional life or from founding a namesake organization to back Parkinson’s research. He has even made the tremors and gait abnormalities characteristic of this common nervous system disorder work for him in his acting career–for example as the foxy, devious lawyer Louis Canning on that delicious TV series, “The Good Wife”.
But even in the rare case when it can be woven into a professional life, living with Parkinson’s disease is not a “fun fact”—although a fun fact is what the E! Entertainment network carelessly called Fox’s Parkinson’s diagnosis during Sunday night’s livestream of the Golden Globe awards. The tremors and behavioral symptoms and inevitable decline that accompany brain depletion of the neurotransmitter dopamine, the hallmark of Parkinson’s, is the very definition of not fun. Not fun for patients, not fun for their families. There are a great many such people. Parkinson’s is the second most common neurological disorder (after Alzheimer’s); patients are estimated to number about 5 million worldwide.
There are, to be sure, therapies like L-dopa that can work reasonably well to restore normal movement for a few years. But these meds have their own burdens, and ultimately the trajectory is down. So researchers have for a long time hunted for more permanent ways of supplying patients with the dopamine they need for normal movement. Last Friday the British medical journal The Lancet published a study showing some promise for one potential approach: Gene therapy, insertion of therapeutic DNA into the brain. The hope is that gene therapy might be a permanent repair.
The ProSavin approach to curing Parkinson’s disease
This particular therapy, called ProSavin, delivers three genes into the brain region that controls movement. The genes code for enzymes that make dopamine. The point of ProSavin is not to repair the damaged dopamine neurons that are no longer producing the neurotransmitter essential for normal movement and other critical functions. Instead the aim is to reprogram neurons that normally don’t produce dopamine, converting them into neurons that do as a replacement for the constant source of dopamine lost in Parkinson’s disease.
The results of the trial, administered to 15 patients with advanced Parkinson’s disease, were encouraging. Motor function–tremors, speech, rigidity, slow movement–improved in all patients at both 6 months and a year after the surgery that delivered the genes. Side effects were mostly mild. The researchers caution against too much optimism, however. The study was small and they noted that the positive effects fall within the range of positive placebo effects seen in studies of other potential therapies for Parkinson’s disease that involve surgery. The work was funded by OxfordBiomedica, a pharmaceutical company in England.
The long, long road to gene therapy
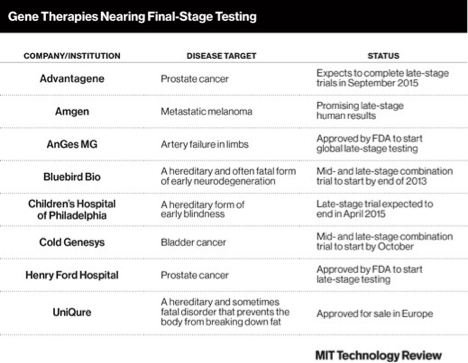
Few gene therapies are available anywhere and none has yet gotten regulatory agency approval in the US. Susan Young of Tech Review says some may be released for sale in the next few years. She presents a short table listing gene therapies that are in final testing stages in the US. They are aimed at diseases that include prostate cancer, melanoma, artery failure, early blindness, early neurodegeneration, bladder cancer and a hereditary lipid disorder.
The Lancet paper is the most recent example of the slow and painful rehabilitation of gene therapy that has been increasingly evident in the past year or so. The idea of curing disease by giving patients the genes they need for normal function began in the 1990s with high hopes, but crashed and burned after the death of patient Jesse Gelsinger in a clinical trial in 1999. It seemed like the end of gene therapy. Carl Zimmer described this sorry history and the potential renaissance of gene therapy in Wired last summer.
In principle it seems like a simple idea for curing disease: if a gene in a cell isn’t working, deliver a functioning gene and let it take over. In practice, of course, not so easy, starting with the problem of gene delivery. How the devil do you get the good gene into the cell and then get it to produce the protein the patient needs?
The plan has been to let the good gene ride into the cell on a virus, since what viruses do for a living is slip into cells and take over their genetic machinery. Of course for that you need a virus that will be talented at delivering its foreign gene cargo but won’t endanger the host.
ProSavin’s virus vehicle of choice is a lentivirus, the first time this particular virus has been used in gene therapy for a nervous system disorder. Lentiviruses are not inclined to stimulate the host immune system, they can carry a large payload, and they are efficient at getting into cells. Lentiviruses are retroviruses, a viral family that includes some nasty customers like HIV. But harmless lentiviruses are widely used in research because they’re such a good gene transport system. They’ve been employed as gene-delivery vehicles–researchers call them vectors–in more than 300 clinical trials.
Another trial of gene therapy for Parkinson’s disease
The National Institutes of Health has also funded a US trial of gene therapy for Parkinson’s disease. The NIH trial uses a different approach from ProSavin, and a different virus too. That’s adeno-associated virus (AAV), a more common vector for gene therapy than lentivirus. Zimmer’s piece will fill you in on the history of research on AAVs as gene therapy vectors. There are now more than 300 of them.
The virus in this trial, AAV2, will not be delivering dopamine-producing enzymes like ProSavin. Instead the cargo will be DNA coding for a brain chemical called glial cell line-derived neurotrophic factor (GDNF). GDNF promotes survival of embryonic dopamine-producing neurons in the lab and has also been beneficial in animal models of Parkinson’s. The researchers will be using AAV2 to deliver human GDNF complementary DNA to brain areas involved in preparing for and controlling movement.
This is a Phase 1 trial, one that aims to investigate whether the gene therapy approach is safe. It is now recruiting patients and is scheduled to complete in 2018. If the therapy proves to be safe, there may be a Phase 2 trial to see if it works to ameliorate Parkinson’s symptoms.
In short, even if everything goes perfectly in the studies of GDNF for Parkinson’s disease, this therapy is at least ten years away, probably more. Gene therapy seems to be climbing back from what looked like oblivion, but the climb is slow and arduous. ProSavin, which last Friday was described as looking promising but is still not ready for Parkinson’s patients, has been in the works for 16 years.
Tabitha M. Powledge is a long-time science journalist and a regular contributor to the Genetic Literacy Project. She writes On Science Blogs for the PLOS Blogs Network. New posts on Fridays.































