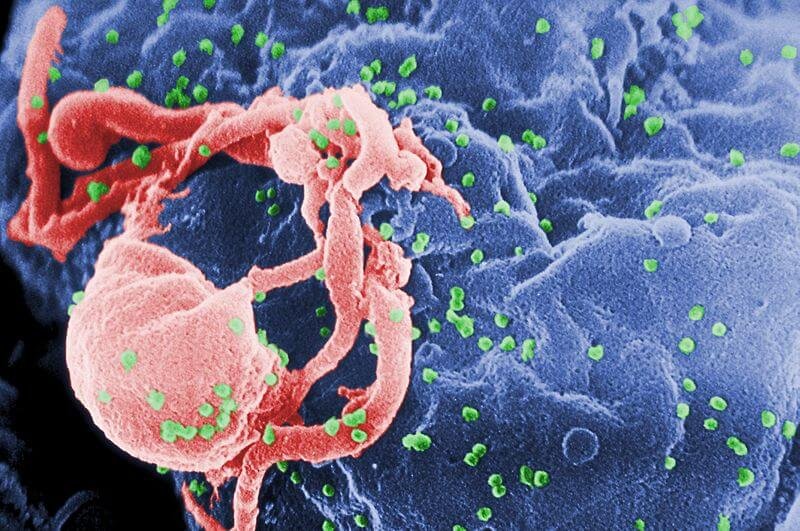
In the three decades that we’ve been grappling with HIV and AIDS. Although we’ve made much progress in managing it we’ve yet to defeat it. Despite a few notable successes, there is no reliable cure or anything resembling a vaccine.
But a pilot study in The New England Journal of Medicine suggests renewed reason for optimism: scientists believe that applying modern gene-editing techniques to our own cells might be the key to finally doing more than simply managing the virus with side-effect laden medications.
A team led by Carl June and Pablo Tebas, immunologists at the University of Pennsylvania in Philadelphia, took inspiration from the story of the “Berlin patient” Timothy Brown who was apparently (and accidentally) cured after bone marrow transplants in 2007 and 2008. He got phenomenally lucky with what was meant as a treatment for his leukemia, not HIV. His donor happened to have two mutated copies of the CCR5 gene, which normally instructs immune cells to produce a protein that HIV latches onto. The mutated copies do not produce the protein; the virus cannot get a foothold.
People with two copies of the mutated CCR5 gene are highly resistant to HIV, and it appears Brown, now producing white blood cells from the donor marrow, developed sufficient resistance to the virus to be cured.
Unfortunately, bone-marrow transplants from HIV resistant patients are not a viable treatment route: there are too few people with the requisite mutations, and the procedure itself is too expensive and risky. But what if we could induce the mutation ourselves, without the risks of an extensive transplant?
That’s exactly what June and Tebas did with a dozen HIV positive patients. They drew blood, cultured it, and used a commercially available gene-editing enzyme called zinc-finger nucleases (ZFNs) to target and disrupt the CCR5 gene in their cells — effectively gifting these cells with the same mutation that gave Brown his cure. They then gave the patients’ their own modified blood back via transfusion.
In short, it worked. Nature summarizes the promising results:
After treatment, all had elevated levels of T cells [white blood cells] in their blood, suggesting that the virus was less capable of destroying them.
Six of the 12 participants then stopped their antiretroviral drug therapy, while the team monitored their levels of virus and T cells. Their HIV levels rebounded more slowly than normal, and their T-cell levels remained high for weeks. In short, the presence of HIV seemed to drive the modified immune cells, which lacked a functional CCR5 gene, to proliferate in the body.
It’s important to remember, as a New York Times report emphasizes early on, that this small study was meant “to test safety, not efficacy.” In that regard it was also a success. Only one patient experienced side-effects: “Jay Johnson, 53, who lives in Philadelphia, took part in the study and was treated in 2010. He was the only patient who had an adverse reaction: a brief bout with fever, chills and joint and back pain.” Johnson also told the Times that he’d gladly go through the treatment again.
It’s important not to discount the effectiveness of modern drugs at keeping the infection at bay — or even, under the right circumstances, curing it.
There are now two cases of HIV being defeated in newborns with early and aggressive antiretroviral treatment. In March of 2013, doctors successfully cured an HIV positive baby using an aggressive and early treatment of antiretroviral drugs. Just last week, doctors in Los Angeles made headlines by reporting that they may have cured a second child of HIV with similar treatment. In both cases, however, the fact that the children were newborns seems essential to their treatment working: they don’t seem to point toward a successful treatment for adults.
For many adults, though, many are able to manage the virus with a few pills a day.
But these are not cures. We know how the famous Berlin Patient was cured, now we have the tools to replicate his unique circumstances through genetic intervention. It’s hard not to see this pilot study as a symbolic achievement for genetic medicine
June and Tebas have taken some basics of biology and reasoned: Viruses need proteins to attach to cells to infect them. Genes tell cells what proteins to produce. Get rid of the protein, get rid of the virus’s access point. Until recently, such a line of reasoning might have gone nowhere, but we now live in a world of increasingly powerful gene-editing techniques. Their preliminary success is the success of the core ideas of personalized medicine.
Kenrick Vezina is the Gene-ius Editor for the Genetic Literacy Project and a freelance science writer, educator, and amateur naturalist based in the Greater Boston area.
Additional Resources: