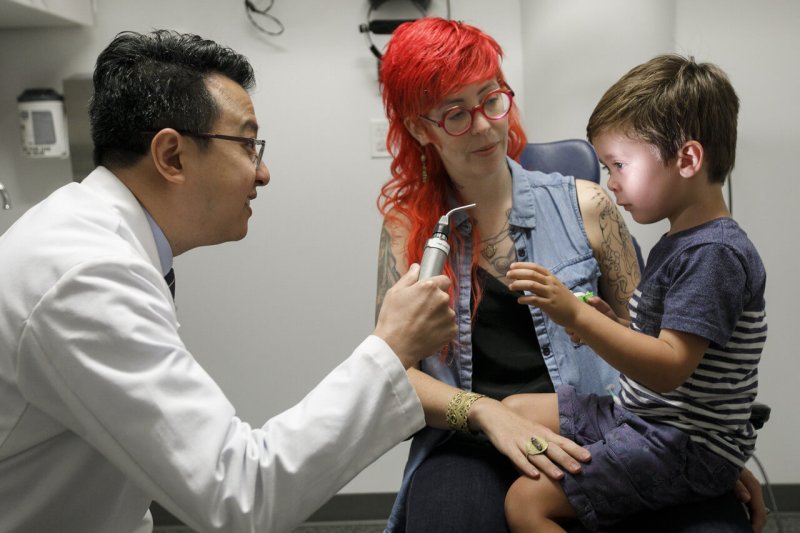
This month Caspian became among the first patients in the country to receive a new gene-therapy treatment called Luxturna.
…
The therapy entails inserting a functional gene through harmless, virus-like particles into the retinal cells to compensate for the faulty gene causing his disease.
It’s among the most expensive treatments on the market, with a price tag of $850,000 to treat both eyes, raising questions about how the health system will absorb such treatments as they become more common.
…
The Institute for Clinical and Economic Review, an independent nonprofit that conducts cost-effectiveness analyses on new therapies, concluded in a February analysis that Luxturna’s price exceeds commonly cited thresholds for cost-effectiveness, in part because its long-term benefits remain unknown.
…
Spark is offering a rebate of up to 20% if the therapy doesn’t work in 30 to 90 days and again 2½ years later, [Spark CEO Jeff] Marrazzo says. It also is offering to pay patient out-of-pocket costs, including copayments. “Of course we’re going to continue to push to get patients more access, but we’re pleased with the start,” he says.
Editor’s note: Full text behind paywall
Read full, original post: High Hopes for a Gene Therapy Come With Fears Over Cost