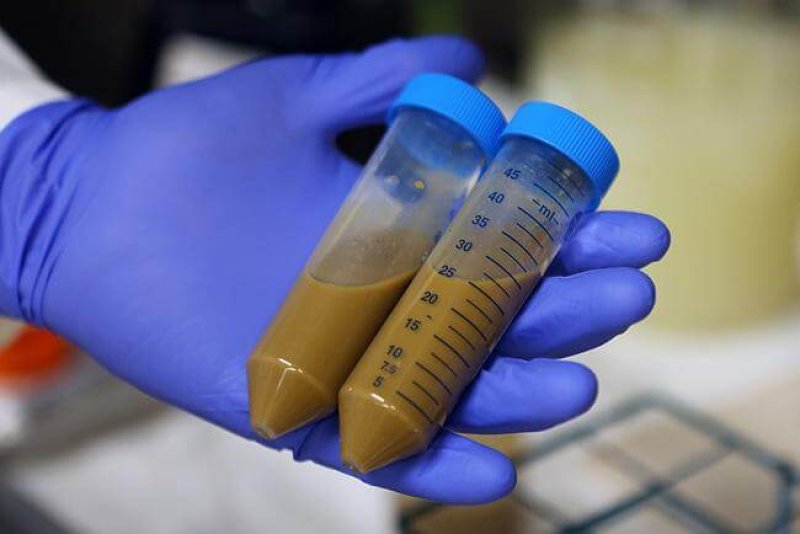
For the past several years, the U.S. Food and Drug Administration has been trying to figure out how to regulate human feces.
Fecal transplants are an almost miraculously effective cure for a gut infection called C. diff. The microbes in the stool of a healthy donor repopulate the gut microbiome of a sick patient. But some of those microbes could be dangerous. [June 13], hypothetical concerns became real when the FDA issued a warning: An immunocompromised patient died after acquiring drug-resistant bacteria from a stool donor.
The death was the first of its kind ever recorded, out of tens of thousands of fecal transplants, and it could very well become a flash point in the battle over regulation. “This is an unintended consequence that the FDA’s really been waiting for, because they’ve had concerns for a long time,” said Colleen Kraft, a microbiologist at Emory University.
…
The FDA is expected to issue its final guidance on fecal transplants very soon. The decision will clear up whether the agency considers fecal transplants to be like drugs; human tissue, such as blood and organs; or an altogether new category of its own.
Read full, original post: Should Human Feces Be Regulated Like a Drug?