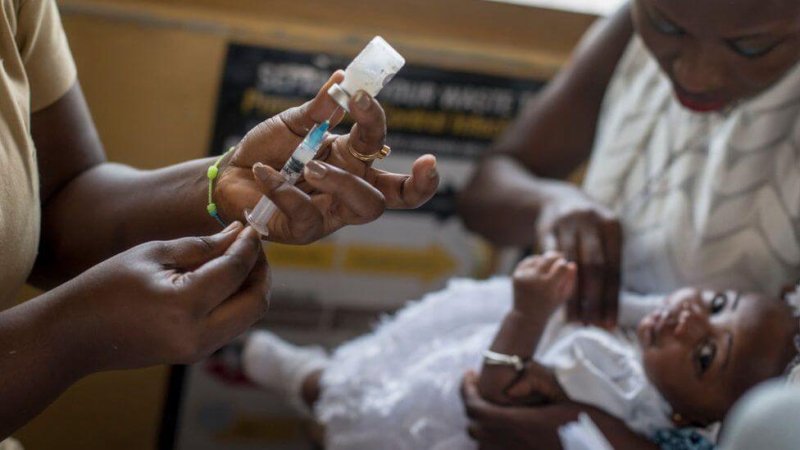
Researchers and health officials have long sought a vaccine against malaria, which in 2019 sickened an estimated 229 million people and caused 409,000 deaths globally. Once infected, many people suffer recurring bouts that undermine their health and quality of life. Pregnant women, children and travelers with no previous exposure to the malaria parasites transmitted by mosquitoes are at highest risk of severe disease.
Since United States military forces are also at risk when deployed to areas where malaria is endemic, its control has long been a priority for the Department of Defense. The novel vaccine based on mRNA technology was developed by scientists from the Walter Reed Army Institute of Research (WRAIR) and Naval Medical Research Center, in partnership with researchers from the University of Pennsylvania and Acuitas Therapeutics.
Researchers used mRNA technology to develop the BioNTech/Pfizer and Moderna vaccines that are highly effective against COVID-19, a feat that malaria researchers noted when publishing their findings in npj Vaccines.
“Recent successes with vaccines against COVID-19 highlight the advantages of mRNA-based platforms — notably, highly targeted design, flexible and rapid manufacturing and ability to promote strong immune responses in a manner not yet explored,” wrote Dr. Evelina Angov, a researcher at WRAIR’s Malaria Biologics Branch and senior author on the paper. “Our goal is to translate those advances to a safe, effective vaccine against malaria.”

WRAIR was previously involved in developing RTS,S, a first-generation product that is currently the most advanced malaria vaccine. RTS,S is based on the circumsporozoite protein of Plasmodium falciparum, the most dangerous and widespread species of malaria parasite. While RTS,S is an impactful countermeasure in the fight against malaria, field studies have revealed limits to its efficacy and duration of protection. So scientists turned to new platforms and second-generation approaches for malaria vaccines.
The mRNA vaccines are not hugely different from how traditional vaccines work. But instead of injecting a weakened live or killed virus, the mRNA approach trains the immune system directly with a single protein. The novel malaria vaccine relies on the circumsporozoite protein to elicit an immune response, similar to RTS,S. However, rather than administering a version of the protein directly, the new approach uses mRNA, accompanied by a lipid nanoparticle that protects from premature degradation and helps stimulate the immune system, to prompt cells to code for circumsporozoite protein themselves. Those proteins, which cannot actually cause infection, then trigger a protective response against malaria.
“Our vaccine achieved high levels of protection against malaria infection in mice,” wrote Katherine Mallory, lead author on the paper. “While more work remains before clinical testing, these results are an encouraging sign that an effective, mRNA-based malaria vaccine is achievable.”
Various methods have been explored in an attempt to reduce malarial infections, including using insecticides to control the mosquitoes that spread the disease, antimalaria drugs, vaccines and barriers like sleeping nets. But they all have their limitations, including the problem of insects developing resistance to pesticides.
The newest efforts under investigation focus on biocontrol strategies, such as using CRISPR to alter a mosquito’s gut genes, employing gene drives that limit reproduction in mosquitoes that carry the disease, applying a fungus that has been genetically engineered to express a spider venom to sheets that can be hung in homes and artificially creating mosquitoes that carry the Wolbachia bacteria, which has been shown to reduce transmission of other mosquito-borne diseases.
It’s likely that a variety of tools will be needed to make a dent in malaria, which remains a serious health concern in sub-Saharan Africa, South Asia, New Guinea and other tropical regions. International researchers fear the disease could become even more prevalent as climate change causes global temperatures to rise, increasing its virulence in areas where it is already endemic and introducing it to regions where it was previously not found.
Joan Conrow has more than 35 years of experience as a journalist and editor. She specializes in environmental issues, biotechnology, and agriculture, and is especially interested in how these highly charged topics are playing out globally. Joan holds a BA in history and journalism and is certified in beekeeping, mediation, and facilitation. Find Joan on Twitter @joanconrow
A version of this article was originally posted at the Cornell Alliance for Science and has been reposted here with permission. The Cornell Alliance for Science can be found on Twitter @ScienceAlly