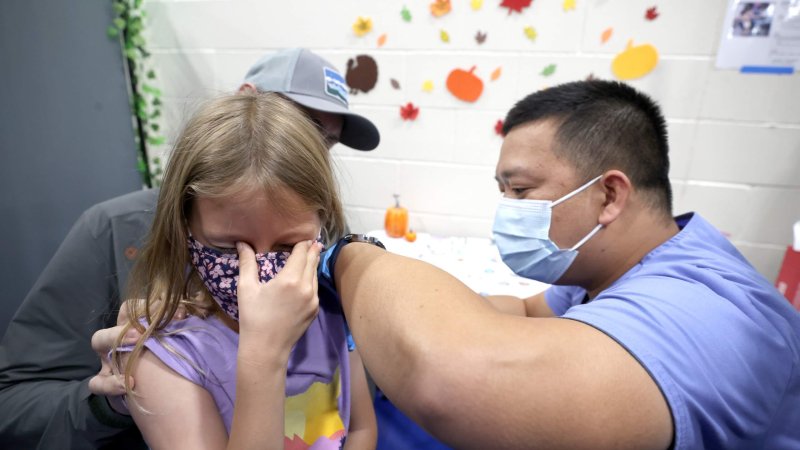
[Soon,] scientific advisers to the [FDA] will decide whether to endorse two doses of the vaccine for children 6 months through 4 years of age, before clinical trials have shown the full course — three doses — to be effective. Such an authorization would be a first for the agency, many experts say.
In fact, interim results from the trial suggested that two doses of the vaccine did not produce a strong immune response in children aged 2 through 4. Results from trials of the third dose are expected in a few weeks.
Although children generally do not become seriously ill when infected with the coronavirus, more of them have been hospitalized during the Omicron surge than at any other point in the pandemic.
But multiple studies have shown that children who are hospitalized with Covid tend to have conditions that predispose them to severe illness, including diabetes, chronic lung disease or heart problems.
Instead of authorizing the vaccine for all 18 million children aged 6 months to 4, the agency might consider recommending it only for children at high risk until more evidence becomes available, some experts said.
…
“On one side, parents are desperate to get their kids protected. On another side, there is extreme distrust,” said Natalie Dean, a biostatistician at Emory University in Atlanta. “The whole process will need to be approached with care and a lot of transparency.”