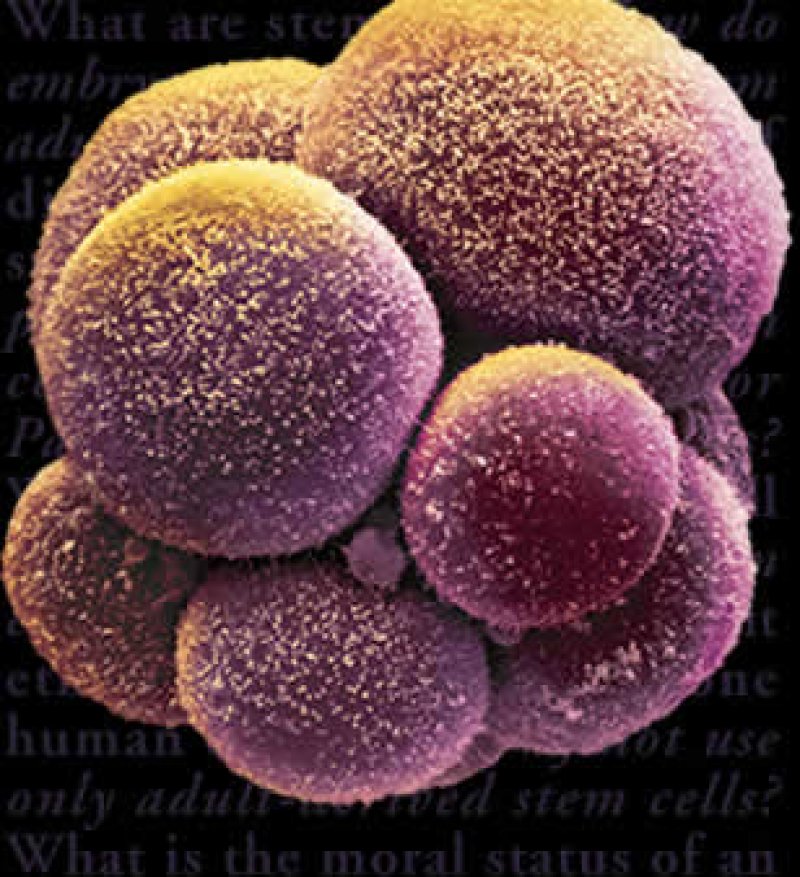
 It’s official: stem cells are drugs. At least, that’s the opinion of the US district court in Washington DC, which has ruled that the Food and Drug Administration (FDA) has the authority to regulate clinics offering controversial stem cell therapies.
It’s official: stem cells are drugs. At least, that’s the opinion of the US district court in Washington DC, which has ruled that the Food and Drug Administration (FDA) has the authority to regulate clinics offering controversial stem cell therapies.
Treatments in which stem cells are harvested from bone marrow and injected straight back into the same patient are deemed part of routine medical practice – not regulated by the US government. But if the cells are subjected to more than “minimal manipulation”, the FDA maintains that the therapy becomes a “drug”, which must be specifically approved for use.
It was on this basis that in 2008 the FDA began moves to shut down Regenerative Sciences, a clinic in Broomfield, Colorado, that treats orthopaedic problems using a stem cell therapy called Regenexx.
View the original article here: Ruling frees FDA to crack down on stem cell clinics