The first genetically engineered animal approved for consumption could be on your dinner table within two years if anti-GMO groups do not succeed in their attempts to block it.
After languishing in regulatory purgatory for almost 20 years, an Atlantic salmon whose genes have been tweaked to grow twice as fast and on less food as farm-raised conventional varieties, was approved by the Food and Drug Administration.
“The FDA has thoroughly analyzed and evaluated the data and information submitted by AquaBounty Technologies regarding AquAdvantage Salmon and determined that they have met the regulatory requirements for approval, including that food from the fish is safe to eat,” said Bernadette Dunham, D.V.M., Ph.D., director of the FDA’s Center for Veterinary Medicine, in a statement.
“I am thrilled by the FDA action, and look forward to bringing AquAdvanage salmon to the market, The approval is a milestone for American science, and I believe will introduce the American consumer to a safe and sustainable salmon food product,” Ronald Stotish, AquaBounty CEO, told me. The company issued a news news release, saying the fish is grown in an “environmentally responsible manner without damaging the ocean and other marine habitats.”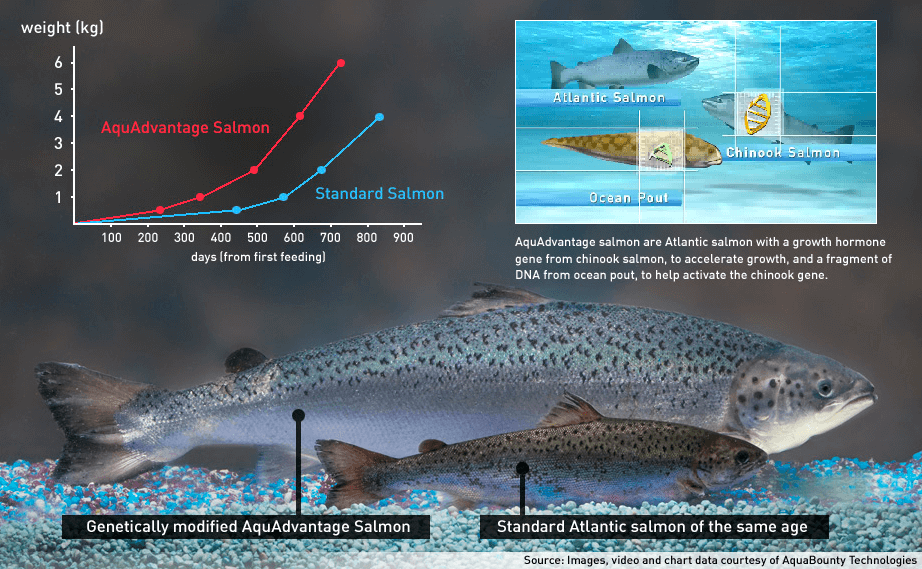
AquaBounty, producer of the AquAdvantage salmon, told me it will not put a special label designating it as genetically engineered. “We’d like to label it as a premium product, but we’ll probably introduce it as ‘Atlantic salmon,’” Stotish said. In its statement, the FDA also announced draft guidance on the voluntary labelling of food derived from the product.
The approval was welcome by most mainstream scientists but kicked off a firestorm of criticism by longtime critics of agricultural biotechnology, who had swamped the Internet over the years with cartoonish  scare stories and pictures of an invasion of Frankenfish.
scare stories and pictures of an invasion of Frankenfish.
“I view the announcement of the FDA to approve the AquaBounty GE salmon for production in land based system as a huge win-win for the environment, consumers, and the process!” William Muir, an animal genetics professor at Purdue University, told GLP sister organization GENeS (Genetic Expert News Service). “The scientific review is clear, there is no credible evidence that these fish are a risk to either human health or the environment.”
Wenonah Hauter of Food & Water Watch denounced the approval as “unfortunate,” saying it came “despite insufficient safety testing and widespread opposition.” FWW and other NGOs called on
President Obama to overturn the decision and said they will be examining all legal options to “stop this unnecessary fish from reaching the marketplace.”
The Center for Food Safety immediately announced it was assembling a coalition of anti-GMO groups to file suit in an attempt to derail the approval.
“The fallout from this decision will have enormous impact on the environment. Center for Food Safety has no choice but to file suit to stop the introduction of this dangerous contaminant,” said Andrew Kimbrell, executive director of Center for Food Safety. “FDA has neglected its responsibility to protect the public.”
Political logjam broken
The green light comes almost three years after the Genetic Literacy Project’s Jon Entine revealed in an investigation in Slate that the approval process had been brought to a halt after political intervention from the White House. That report embarrassed the White House into lifting its hold on the approval process the afternoon after the story was posted. That led the FDA to publish its positive environmental assessment in late December 2012, and ask for public input, a process expected to be concluded within months. But reports surfaced that the White House again intervened, leading to an additional three-year delay.
Meanwhile, in 2013, the Canadian government issued its evaluation of the GE salmon, describing its risk to the Canadian environment from AquaBounty’s Prince Edward Island growing facility as “negligible” and the risk to human health as “low.”
It’s not clear what broke the political logjam. Over the past three years, sources within the FDA have repeatedly told me that the scientific review process is complete and the agency was not the source of the holdup. Independent scientists have been critical at the delay.
“This shouldn’t be happening,” said Gregory Jaffe, director of biotechnology at the Center for Science in the Public Interest, while the process dragged on. Jaffe had participated in a scientific review panel in 2010 that unanimously endorsed the FDA’s findings in 2010 that the salmon was safe to eat and for the environment. “AquaBounty deserves regulatory due process,” he added. “We need science-based decisions made in a timely fashion. The public deserves this, and there are questions whether that is what’s going on in this case.”
The GE fish, developed by AquaBounty, now majority owned by Intrexon Corporation, is an Atlantic salmon with two added genetic elements: a Chinook salmon growth hormone gene and an on-off switch from the ocean pout, another edible fish. Test versions of the salmon have been grown in two inland facilities, in Canada and Panama
Americans consume 650 million pounds of salmon each year, with more than 530 million pounds of that imported. When it finally reaches the marketplace, the GE salmon could lead to lower salmon prices and an increase in consumption of salmon, a heart-healthy food rich in protein and other nutrients. It offers numerous sustainability advantages, say its proponents. It was developed using a land-based closed-containment systems that conserves water and allows production without antibiotics or other chemicals to control disease and parasites.
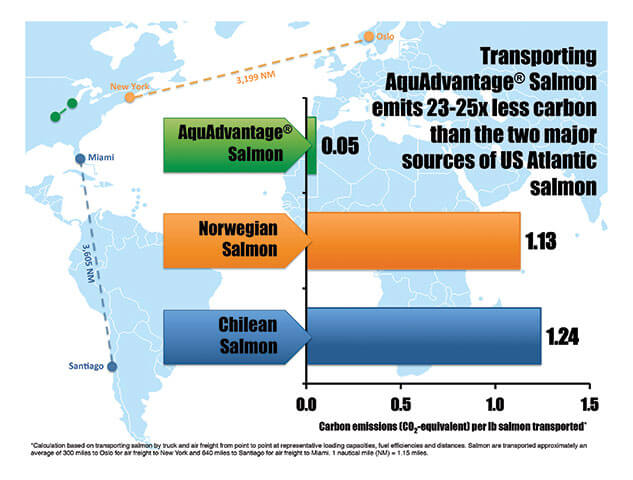
The salmon consumes about 25% less feed to reach full size. The US currently imports over 90% of all of its seafood, and specifically 95% of the Atlantic salmon, it consumes. Stotish noted that because the salmon can be farmed close to major consumer markets, it will cut down on greenhouse gas emissions from flying in imported varieties and the environmental impacts associated with current salmon farming operations.
Winding road
AquaBounty initiated its application to commercialize in 1995. GM crops and animals are regulated under the 1986 Coordinated Framework. But while plants have a clear path to approval under guidance in place by 1992, animals must travel through regulatory no-man’s land. Until the approval of the GM salmon, the FDA has approved only one product, an anticoagulant derived from the milk of transgenic goats.
By 2004, AquaBounty had assembled its “data package,” but its path to approval was never entirely clear. Finally, in 2008, the Bush administration decided that transgenic animals intended for the dinner table would be regulated as animal drugs by the FDA’s Center for Veterinary Medicine.
Anticipating environmental concerns, AquaBounty evaluated whether the fish performed well under standard commercial conditions at its Canadian and Panamanian facilities. As part of its evaluation, the FDA inspected the Panama site, determining the fish would be securely contained with multiple redundant systems that would prevent the salmon from escaping into the wild—one of the main concerns for people opposed to GMOs. The FDA concluded that even a catastrophic event would not pose a threat. Lengthy expanses of warm, muddy water outside the facility would serve as a graveyard to any escaped cold-water fish. If some somehow made it to the ocean, they would die in the warm currents thousands of miles from their spawning grounds in the frigid waters of the North Atlantic.
There is no chance, independent scientists say, that released salmon would win a Darwinian war in open waters with wild salmon—the so-called Trojan gene effect. GMO opponents cite a 1999 study concluding that modified fish that grow extra large would have a competitive advantage, threatening extinction of conventional varieties. But AquaBounty engineered the salmon so it grows no larger than conventional fish. A co-author of that study, Purdue’s Bill Muir, who developed the risk assessment model for transgenic fish for the Department of Agriculture, studied the AquaBounty salmon and determined it has no fitness advantage—and now endorses it.
“The current practice of using wild caught salmon as a food source is not sustainable, our oceans are over fished, said Purdue’s Muir. “This development provides a safe and sustainable alternative. Further, this opens the door for the use of biotechnology as a tool to improve US food production in other species and in other ways to help feed a growing population.”
Friends of the Earth, Center for Food Safety, Greenpeace, Union of Concerned Scientists, and other anti-GMO groups have been steadfastly critical off the first animal GMO, and have been furiously  lobbying to prevent the final approval. They claim the approval disregarded AquaBounty’s environmental record, they claim has raised the stakes for an environmentally damaging escape of GMO salmon. Consumers Union, an ardent opponent of GM foods, said in 2013 that the FDA’s determination that escape was a remote possibility was built on “inadequate science and unfounded assumptions.”
lobbying to prevent the final approval. They claim the approval disregarded AquaBounty’s environmental record, they claim has raised the stakes for an environmentally damaging escape of GMO salmon. Consumers Union, an ardent opponent of GM foods, said in 2013 that the FDA’s determination that escape was a remote possibility was built on “inadequate science and unfounded assumptions.”
F&W said that AquaBounty facilities outside the U.S. have dealt with an accidental disease outbreak, an accident that lead to “lost” salmon, and a $9,500 fine from Panamanian regulators who found the company in breach of that country’s environmental laws.
The fish has also been in the political crosshairs, with House and Senate members from key salmon producing stakes publicly opposing the approval, fearful that it could harm their states economies. “Frankenfish is uncertain and unnecessary,” said Alaska Republican Don Young in 2011, who emerged as a spokesman for more than a dozen House members. “Should it receive approval as an animal drug, it clears the path to introduce it into the food supply.”
Future of animal biotechnology
The interminable delay has all but gutted animal biotechnology research efforts in the United States. China has launched an $800 million public-private investment into transgenic animals, and genetically modified animals are being developed in India, New Zealand, and across Latin America, including in Cuba. But North America has become a dead zone.
James Murray, an animal scientist at the University of California–Davis has developed goats that make milk with diarrhea-preventing lysozyme, a bacteria-fighting protein that could save children’s lives. With no government or private money on the horizon, he’s set up his lab in Brazil, a more biotech-friendly locale.
“When you don’t have a regulatory pathway forward and the government doesn’t support research in this area, what company will invest in this field?” he said to me. “None. The AquaBounty situation is just confirmation of a hopelessly politicized process.”
The future of animal genetics is so dire, universities have been killing off courses. “My program started off doing genetic engineering,” Alison Van Eenennaam, a University of California–Davis animal scientist who co-authored a scathing article for Nature Biotechnology on the broken approval process, told me. “I couldn’t get any government funding for my work in this area, so I shut the program down. Why would I train graduate students for jobs that won’t exist?”
Van Eenennaam hopes that this approval could help remove what she believes is an unfair stigma attached to one animal breeding method, precise genetic engineering. The AquAdvantage salmon was originally developed as a fast-growing variety of salmon by a group of Canadian public university scientists over a quarter of a century ago,” she noted. “It is not unusual for breeders to select for fast-growing, productive plants and animals. Unique to animals bred using one breeding method (genetic engineering), however, is a lengthy and expensive regulatory process.”
“The significance of the action is that it marks the first approval globally for production of genetically modified animals for purposes of food production and sale,” said Eric Hallerman, Professor of Fish Conservation at Virginia Tech University.
Other scientists supportive of the approval remain cautious and urge continued oversight. Greg Jaffe:
[The Center for Science in the Public Interest] agrees that the extremely limited production allowed in today’s action will not harm the environment, and may even be beneficial if AquaBounty’s claims of efficiency are realized. To the extent that AquaBounty asks FDA for additional production capacity in other parts of the world, including the United States, the agency should conduct a new environmental assessment and impose conditions to ensure that the salmon could not escape and alter the surrounding ecosystems.
Whether consumers will embrace the world’s first genetically modified fish remains to be seen. As Tamar Haspal reported in the Washington Post:
Some retailers have already expressed reservations. A few supermarket chains, including Whole Foods and Trader Joes, have pledged not to sell the salmon, and others, like Safeway and Kroger, have issued less definitive statements indicating that they’re not planning to stock the fish. The 2014 Safeway statement, for example, says that, “we are not considering nor do we have any plans to carry GE salmon.
Now the legal and public relations war is set to escalate dramatically.
Jon Entine, executive director of the Genetic Literacy Project, is a Senior Fellow at the World Food Center’s Institute for Food and Agricultural Literacy, University of California-Davis. Follow @JonEntine on Twitter

































