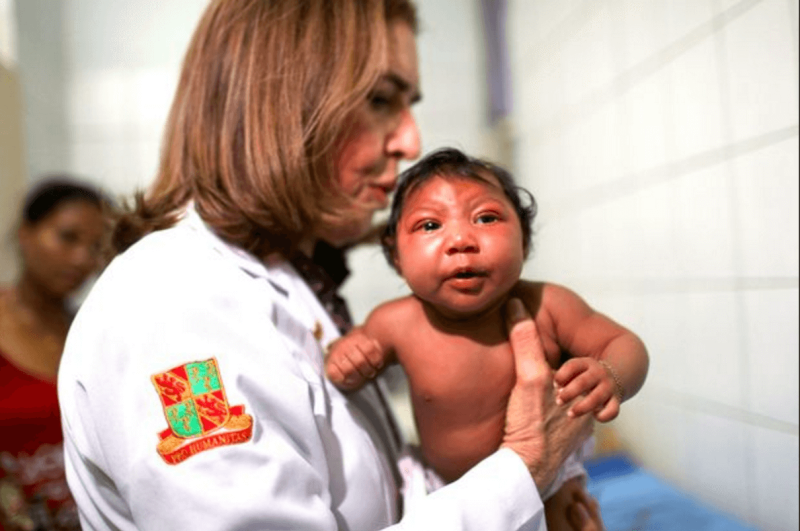
Zika virus has recently transformed from a relatively unknown tropical disease to a worldwide public health emergency. This crisis is due to emerging evidence that Zika causes microcephaly and Guillain-Barré syndrome (GBS), two neuronal disorders with severe symptoms.
By now, it has been widely reported that Zika may be linked to these two disorders, but the process of verifying that connection has been arduous and is still ongoing.
Zika is a flavivirus that was first discovered in Uganda in 1947. Like several other well-known flaviviruses (such as yellow fever, dengue, and Chikungunya), Zika is transmitted to humans primarily by the Aedes aegypti and the Aedes albopictus mosquitoes, whose habitat has slowly expanded to cover every continent except Europe and Antarctica. The spread of these mosquitos has precipitated several flavivirus outbreaks in the Americas, with Zika being the most recent.
In May 2015, Brazilian public health officials noticed an outbreak of rash and fever in the northeast of the country. These patients tested negative for the most likely causes of those symptoms, but 16 tested positive for Zika. By the end of June, the number of Zika cases had increased to more than 14,000.
Six months later, the Brazilian Ministry of Health (SUS) and Pan-American Health Organization (PAHO) noted a 20-fold increase in cases of babies born with microcephaly in the same regions that had seen Zika outbreaks. Microcephaly literally means “small head”, and it is typically defined as a head circumference two standard deviations below the average for the age and sex of the baby before or after she is born. While this diagnosis is technically straightforward, involving one simple measurement, it leads to a very broad disease definition. Many children born with microcephaly develop brain disease, seizures, and developmental disorders, but their symptoms vary in ways that are not completely predictable. Similarly, there are many causes of microcephaly, including specific genetic mutations, drug use, malnutrition, and viral or bacterial infections. Not every infection can be transmitted to the fetus during pregnancy, but a specific set known as TORCH (Toxoplasmosis, “Other” including HIV and syphilis, Rubella, Cytomegalovirus, and Herpes Simplex) can cross the placenta and cause a range of birth defects including microcephaly.
In addition to the geographical overlap, public health officials had a biological reason to suspect Zika as a new TORCH infection, even with limited data. Only 14 cases of Zika had been reported before 2007, but once Zika expanded beyond its native habitat in East Africa and Southeast Asia, almost 10,000 cases were confirmed in Oceania between 2007 and 2015. In public health terms, 10,000 cases was not enough to draw broad conclusions about the effects of Zika, but in two cases, doctors found Zika in the blood of 4-day-old newborns whose mothers had been infected with the virus. Transmission from mother to fetus is the first criterion for a TORCH infection, and this possibility prompted the Brazilian SUS to look for Zika in the reported cases of microcephaly.
To link Zika to microcephaly, scientists first have to make sure that the mother and child have both been infected with Zika. Detecting Zika is a theoretically straightforward, but often technically challenging problem. There are two main techniques for detecting viral infection — immunologic and genomic — and both have pros and cons.
The immunologic approach takes advantage of the fact that the human body fights viruses on its own by making antibodies that specifically recognize and destroy them. If isolated, these antibodies will still recognize viruses outside the body, and this allows scientists to take blood from a patient and combine it with purified virus in a controlled lab environment. If antibodies from the patient’s blood stick to the virus they are said to be reactive — that is, able to recognize the virus — which is a clear sign that the patient has been infected in the past even if they are no longer affected. Doctors typically test for two types of antibodies: IgG and IgM. IgM antibodies are produced immediately after the immune system recognizes a virus, but they last for less than a year. IgG antibodies, on the other hand, remain in the blood for a longer time period to protect against a second infection. Thus, a positive IgG antibody test indicates that someone has been infected at some point in their life, while a positive IgM test denotes a recent infection.
Antibodies recognize specific patterns on the surface of viruses, and this can present a problem for diagnosis of similar infections. On the outside, Zika looks very much like the dengue virus, and many antibodies to dengue virus are “cross-reactive” with Zika — in other words they are able to bind to both viruses with similar strength. This means that a patient previously infected with dengue could test positive on a Zika immunologic test, and this false positive result could prevent scientists from drawing the right conclusions about Zika’s impact.
Genomic evidence for infection, meanwhile, is a smoking gun analagous to DNA evidence in a criminal trial. DNA is unique to each individual living thing on earth, and finding the DNA of a virus in a patient’s blood or tissue is the clearest possible sign that that person has an infection with that specific disease. Zika, though, carries its code of life as RNA. RNA is similar to DNA, but small structural differences make it less stable and more difficult to handle in the lab. Still, a technique called reverse transcriptase polymerase chain reaction (RT-PCR) can be used to convert RNA to readable DNA.
The drawback of RT-PCR is that it detects the virus directly and thus requires an active infection. As the immune system clears the virus from the body, it consequently eliminates the presence of viral RNA. Therefore there is a very short timeline (typically less than a week) for virus to be detected by RT-PCR.
Zika and Guillain-Barré syndrome
Better detection could have more definitively established a link between Zika and neuropathy during the last outbreak in French Polynesia in 2013. That epidemic occurred at the same time as a dengue outbreak, which made separating the two diseases tricky. Shortly after experiencing Zika fever-like symptoms, a woman in Tahiti was diagnosed with GBS, a devastating disorder in which the body’s immune system begins to attack and damage peripheral nerves. Unfortunately, when she was tested by RT-PCR, the woman came back negative for active Zika and Dengue. While the antibody test was positive for a Zika infection, dengue has been shown previously to cause GBS. With the possibility of a cross-reactivity of antibodies, researchers were not completely convinced that Zika could cause GBS.
Like microcephaly, GBS is often caused by infections. Many of the TORCH infections, as well as HIV, Campylobacter, and Epstein-Barr virus, can give rise to GBS. Generally speaking, GBS is thought to be caused by another kind of cross-reactivity, in which antibodies elicited by the infection not only attack the virus but also the body’s own nerve cells, leading to damage throughout the peripheral nervous system. GBS can be deadly due to respiratory or cardiac failure. The Colombian health minister has confirmed three GBS-related deaths in Zika-infected areas since November 2015, providing yet another possible link between Zika and neuronal damage.
Does Zika cause microcephaly?
When the Brazilian SUS released its first report on microcephaly, it had already performed initial tests in two pregnant women whose fetuses were diagnosed with microcephaly by ultrasound. The RT-PCR test for Zika in the women’s amniotic fluid — which surrounds the growing fetus — had come back positive, providing conclusive evidence that the virus was able to cross the placenta. The TORCH infections provide clear precedence for what can happen when a virus is transferred from a pregnant mother to her fetus, and the SUS took these tests as sufficient evidence to declare a public health emergency. What sets Zika apart compared to the TORCH infections is that it is a flavivirus transmitted by mosquitoes. Prior to Zika, no mosquito-borne virus had ever been suspected to cause birth defects.
Over the next two months, more reports of microcephaly and Zika came in, the WHO declared a Public Health Emergency of International Concern, but no new definitive evidence emerged linking Zika to microcephaly. The Centers for Disease Control and Prevention (CDC) in the United States used this time to develop the necessary protocols and reagents for large-scale RT-PCR Zika testing. On February 19, the CDC released a report stating that Zika had been detected by RT-PCR in the brain and placenta of two newborns with microcephaly and two stillborn babies. This evidence now makes it clear that Zika can not only cross the placenta but infect the fetus’s developing brain, providing a possible mechanism for development of microcephaly. On February 22, CDC epidemiologists landed in Brazil to begin a controlled study of 100 microcephaly patients to understand how large a role Zika may have played in those cases.
The recent surge in Zika research has come amidst reports that Brazil’s original microcephaly estimates may not be accurate. While over-reporting may very well be a real phenomenon, the direct evidence for viral RNA in the brains of babies born with microcephaly is hard to ignore given the precedent of TORCH infections and microcephaly. The CDC study will help determine whether the overlap of Zika and microcephaly occurs in individuals rather than just geographic regions. Time will tell whether the observations in Brazil will be consistent with other countries — Colombia has yet to see an increase in cases of microcephaly, although they have reported an increase in GBS.
Future work: Quest for empirical evidence
As the CDC gathers its info, basic science labs have begun to pursue experimental evidence on the effects of Zika. Some virologists have begun testing strains of Zika to see if they can find one capable of infecting mice in order to perform laboratory tests. Others have begun testing the immune reactions of trophoblasts — protective cells that line the developing embryo — to Zika, and others are testing to see whether previous flavivirus infections can make Zika symptoms worse. Together, these studies could provide experimental and analogous evidence for Zika and microcephaly — but it will likely be a long time before the results are known.
For now, the connection between Zika and microcephaly is still tentative, but the WHO is taking a cautious stance. Meanwhile, other groups have begun focusing on Zika prevention, both from standpoint of vaccine development and “vector control” — eliminating the mosquitoes that carry the virus. The Obama administration has requested $1.8 billion from Congress to fund five phases of Zika research: natural history, viral biology, vector control, diagnostics, and a vaccine. Each of these components will be important for moving forward our understanding and control of Zika in the coming months and years.
This article a cross-publication from Science in the News for which it was originally written. You can follow them on Twitter @SITNBoston. Graphics were prepared by Daniel Utter.
Nathan Schauer is a graduate student in Harvard University’s Chemical Biology program. In lab, he makes a lot of glow-in-the-dark molecules and uses them to study the body’s trash sorters.