Isn’t the appendix fairly useless, even dangerous when inflamed? Is it also a gateway to a brain disease?
Actually, a link between the Parkinson’s protein and the appendix has been known for a few years. Now, new experiments reported in Science Translational Medicine confirm the connection and suggest a protective role for appendectomy. But rather than advising people to shed their appendixes, the researchers see their findings as opening up a new target for drug discovery for Parkinson’s.
“We have shown that the appendix is a hub for the accumulation of clumped forms of alpha-synuclein proteins, which are implicated in Parkinson’s disease. This knowledge will be invaluable as we explore new prevention and treatment strategies,” said Bryan Killinger, first author of the report and a post-doctoral researcher in the laboratory of Viviane Labrie at the Center for Neurodegenerative Science, Van Andel Research Institute in Grand Rapids.
The much-maligned appendix is quite active. It produces 71 percent of all of the nearly 20,000 types of human proteins, so it must be doing something. The most abundant 200 proteins also are prominent in the spleen, tonsils and lymph nodes, creating a quartet of body parts that protect us from infection. Plus, the tiny organ regulates the microbiomes of the small and large intestines, serving as a reserve to replenish resident bacteria.
Shape shifters
Alpha-synuclein is one of several proteins that naturally folds into more than one three-dimensional shape, based on chemical attractions and repulsions between their parts. (Imagine how a dropped strand of cooked spaghetti might land in different ways.) Although a single gene tells a cell how to synthesize a single molecule of a protein – a monomer – rare alternate shapes can trigger a stickiness that clumps the monomers into deposits.
The brain is particularly vulnerable to diseases of protein aggregation. Misfolded TDP-43 protein lies behind a familial form of ALS, as do amyloid beta precursor protein and tau proteins for some forms of familial Alzheimer’s disease.
Parkinson’s has a different protein culprit. “Alpha-synuclein is normally present in the tissues of the body in a native state that is referred to as a ‘disordered monomer.’ When alpha-synuclein clumps, forming aggregates, this is associated with the hallmark pathology of Parkinson’s disease, the Lewy bodies, and is neurotoxic in the brain,” Viviane Labrie told Genetic Literacy Project.
The harmless form of alpha-synuclein was known to be in many organs, including the brain and the appendix. “In our study, we found that aggregated forms of alpha-synuclein, the clumped form that is relevant to Parkinson’s disease, were abundant in the human appendix. If aggregated alpha-synuclein protein leaves the GI tract, from the appendix, to go to the brain, we think that this could be a trigger for Parkinson’s disease,” Dr. Labrie said.
Links to the appendix
Alpha-synuclein inhabits a different neighborhood of the pouch-shaped organ than the abundant lymphoid tissue. It accumulates inside the neurons that are part of the meshwork that innervates the gastrointestinal (GI) tract, called the enteric plexus. From there, the vagus nerve sends its axons up to the brainstem.
Might alpha-synuclein occasionally hitch a ride aboard the vagus nerve to the brainstem, and from there, to the substantia nigra, the epicenter of Parkinson’s? An origin in the appendix might explain why gastrointestinal symptoms, particularly constipation, may begin up to 20 years before the motor symptoms of Parkinson’s.
In 2007, a trio of researchers from Essex Neuroscience Centre in the UK proposed a scenario linking the GI tract to the brain: swallowing viruses and bacteria in nasal mucus, which travel along the enteric nerves from the stomach to the brain. But Parkinson’s isn’t an infection.
A study in 2014 found non-clumped alpha-synuclein in the lining of the appendixes in healthy people. That led the researchers from the University of Ottawa to hypothesize that having had an appendectomy might affect Parkinson’s risk.
In 2015, researchers in Denmark added another piece to the puzzle. They found a lower incidence of Parkinson’s among people who’d had their vagus nerves cut to treat ulcers.
Yet other studies found that alpha-synuclein forms deposits in the GI tract years before motor symptoms of Parkinson’s arise. The time frame is critical, because young people tend to have appendectomies, whereas older ones develop Parkinson’s. That’s why it’s important for a study to cover decades of data. Several previous investigations might not have identified an appendectomy-Parkinson’s link because they only went back a few years, the researchers write.
Biochemistry meets epidemiology
The new work investigated two databases. The Swedish National Patient Registry followed all people in Sweden with any diagnoses since 1964, meeting the criterion of time. Of 1,698,000 individuals, 551,647 had appendectomies, and 2,252 of those developed Parkinson’s. That’s a risk 19.3 percent lower than for people who developed the disorder but retained their appendices. And age at diagnosis was on average 1.6 years later among people who’d had appendectomies.
A second database, the Parkinson’s Progression Markers Initiative, covered people diagnosed with the disorder and included the age that symptoms began, demographic factors, and genetic information (most cases of Parkinson’s are not inherited). Of the 849 cases, 54 had had an appendectomy.
Having had an appendectomy 30 or more years before the first symptoms of Parkinson’s was associated with an average delay of onset of 3.6 years. However, the delay didn’t happen for people known to have a Parkinson’s mutation, suggesting that any protective effect of having an appendectomy might apply only to cases associated with exposure to environmental risk factors, such as pesticides used in agriculture.
The severity of Parkinson’s was similar whether or not a person had had an appendectomy. That suggests that the influence of the appendix is to seed the neurological disease, not accelerate it, the researchers wrote.
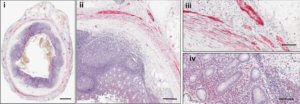
The researchers also probed the appendices of 48 healthy (other than their appendicitis) folks of varied ages, and were surprised to find that 46 of them had lots of the gummy form of alpha-synuclein thought to only be associated with Parkinson’s. The protein was squirreled away in neurons and a few macrophages, the blobby giant scavenger cells of the immune system.
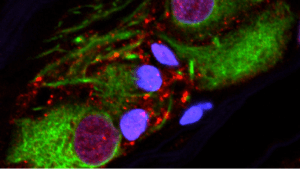
Yet another set of experiments caught the appendix in action, in vitro. The researchers made a soup of sorts out of healthy appendixes and added monomers of the alpha-synuclein manufactured using recombinant DNA technology – that is, outside the body. Sure enough, the appendix soup knit the monomers into a sticky mess, like in a Parkinson’s brain.
“We were surprised that pathogenic forms of alpha-synuclein were so pervasive in the appendixes of people both with and without Parkinson’s. It appears that these aggregates – although toxic when in the brain – are quite normal when in the appendix. This clearly suggests their presence alone cannot be the cause of the disease,” Dr. Labrie said. Something else, perhaps a confluence of events, enables the appendix to raise risk of Parkinson’s, which affects less than 1 percent of the population, she added. “That’s what we plan to look at next: Which factor or factors tip the scale in favor of Parkinson’s?”
Will “prophylactic appendectomy” become a thing? Not likely, because any protection offered by appendix removal isn’t huge. The researchers put it this way in their paper: “In total, PD was diagnosed in 1.17 out of every 1,000 people who had an appendectomy compared to 1.4 per 1000 in the general population.”
While young people should hardly be volunteering up their appendixes to protect against a neurological condition unlikely to show up for decades, the link between the maligned digestive organ and the Parkinson’s brain offers an enticing new drug target.
“Our results point to the appendix as a site of origin for Parkinson’s and provide a path forward for devising new treatment strategies that leverage the gastrointestinal tract’s role in the development of the disease,” Dr. Labrie said.
Ricki Lewis is the GLP’s senior contributing writer focusing on gene therapy and gene editing. She has a PhD in genetics and is a genetic counselor, science writer and author of The Forever Fix: Gene Therapy and the Boy Who Saved It, the only popular book about gene therapy. BIO. Follow her at her website or Twitter @rickilewis













































