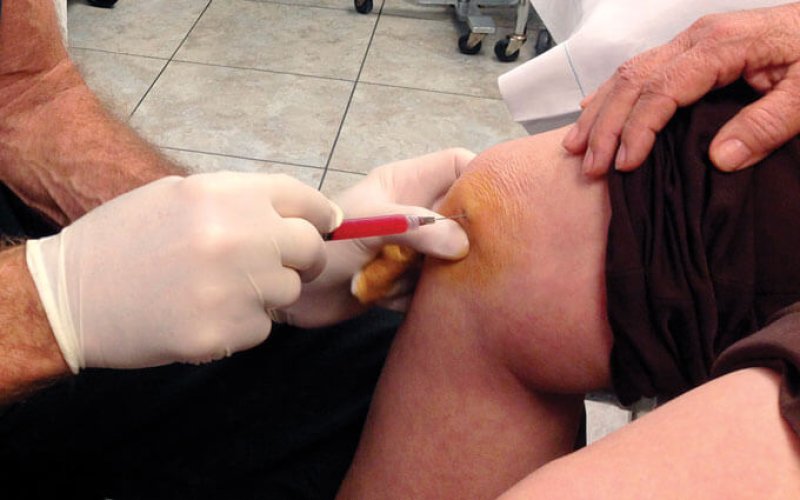
Stem cells sold at clinics are driving what’s thought to be a $2 billion global industry. Facebook pages announce seminars. Local newspapers are wrapped in ads vowing “relief without surgery.” Stem cells are billed as treatments for everything from autism to multiple sclerosis to baldness. Most commonly, the ads focus on orthopedic issues, especially aching knees.
An important point gets left out of the cheery ads: There’s not enough science to justify using stem cells for any of the advertised conditions, including joint pain. None of the treatments advertised have been approved by the U.S. Food and Drug Administration. (The only approved stem cell treatments are for certain cancers and blood disorders.) Very few of the orthopedic studies in humans have been scientifically rigorous, and none have shown stem cells regrowing cartilage.
It’s not even clear whether treatments being touted as “stem cells” contain viable stem cells or whether the contents should be defined as stem cells at all. As the stem cell industry grows rapidly, many researchers who are studying stem cells for their potential to regenerate tissues worry that the booming marketplace, which conflates hype with reality, might ultimately damage research progress.