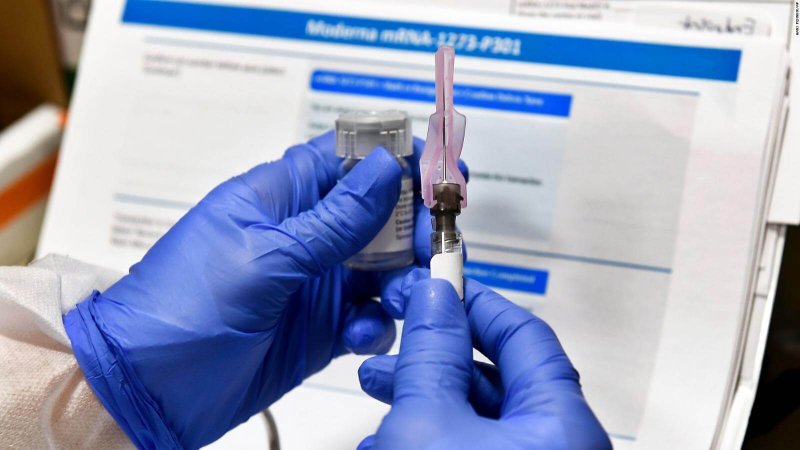
It’s standard for drug companies to withhold details of clinical trials until after they are completed, tenaciously guarding their intellectual property and competitive edge. But these are extraordinary times, and now there is a growing outcry among independent scientists and public health experts who are pushing the companies to be far more open with the public in the midst of a pandemic that has already killed more than 193,000 people in the United States.
These experts say American taxpayers are entitled to know more since the federal government has committed billions of dollars to vaccine research and to buying the vaccines once they’re approved. And greater transparency could also help bolster faltering public confidence in vaccines.
…
“We’ve never had such an important clinical trial — or series of clinical trials — in recent history,” said Dr. Eric Topol, a professor of molecular medicine at Scripps Research in La Jolla, Calif., and a longtime expert on clinical trials. “Everything should be transparent.”