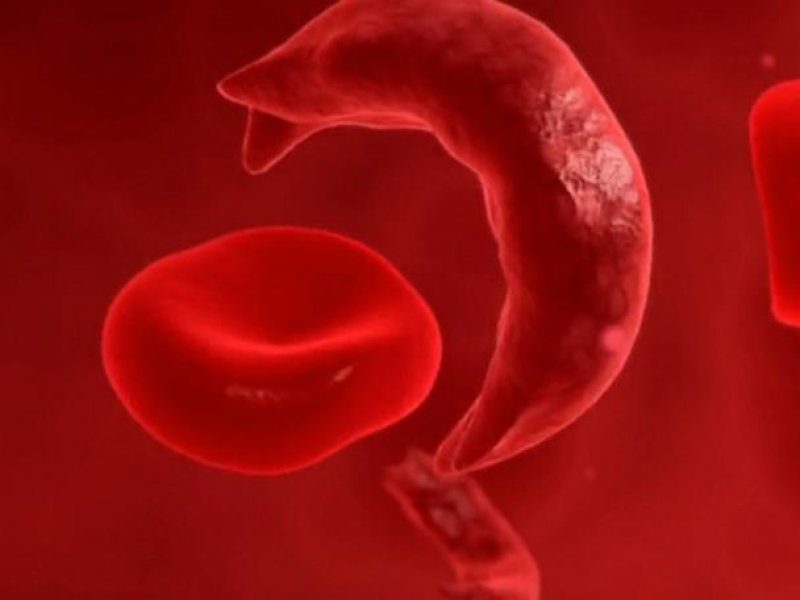
Called Casgevy, the gene-editing treatment is for people with sickle cell disease and a related blood disorder called beta-thalassemia. UK regulators approved the treatment in November 2023, followed by the US and Europe in December. Vertex, the pharmaceutical company that markets Casgevy, announced in a November 5 earnings call that the first person to receive Casgevy outside of a clinical trial was dosed in the third quarter of this year. The company reported $2 million in revenue from that patient. (Casgevy debuted with a price tag of $2.2 million in the US.)
“Cagevy has been enthusiastically received by patients, physicians, and policymakers, and the launch is gathering momentum across all regions,” Stuart Arbuckle, Vertex’s chief operating officer, said on the earnings call. He added that additional patients are accessing the treatment commercially.