And remarkably, that approval was granted in five months, at a time when the average approval time was 31.5 months (similar to today).
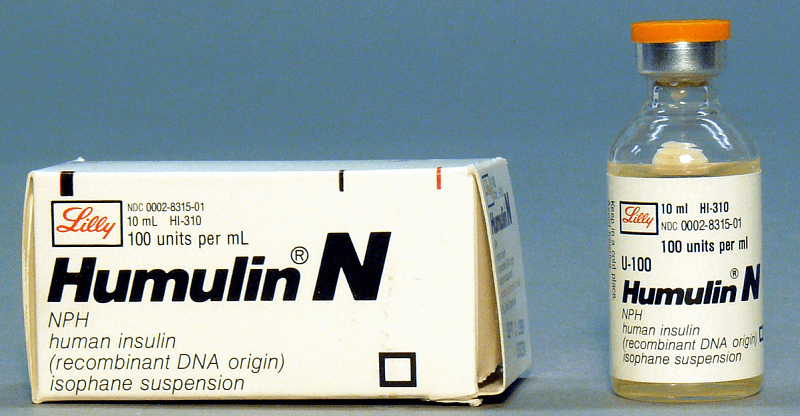
Insulin is secreted in the pancreas and is essential to the metabolism of carbohydrates and fats. Insulin deficiency leads to the development of (Type 1) diabetes, requiring that patients have regular injections of insulin to maintain life and health.
As the FDA medical reviewer in 1982 and the head of the evaluation team for “Humulin,” the brand name of the human insulin, I had a front-row seat. The saga is remarkable in several ways, not least of which is that although both the drugmakers and regulators were exploring unknown territory, the development of the drug and its regulatory review progressed smoothly and rapidly.
Before genetically-engineered insulin
Insulin in crude form was first produced a century ago, in 1922, by Canadian researchers Frederick Banting and Charles Best, which lifted the death sentence that had previously been imposed on diabetics. By the end of that year, drug company Eli Lilly and Company had devised a method for much higher purification.
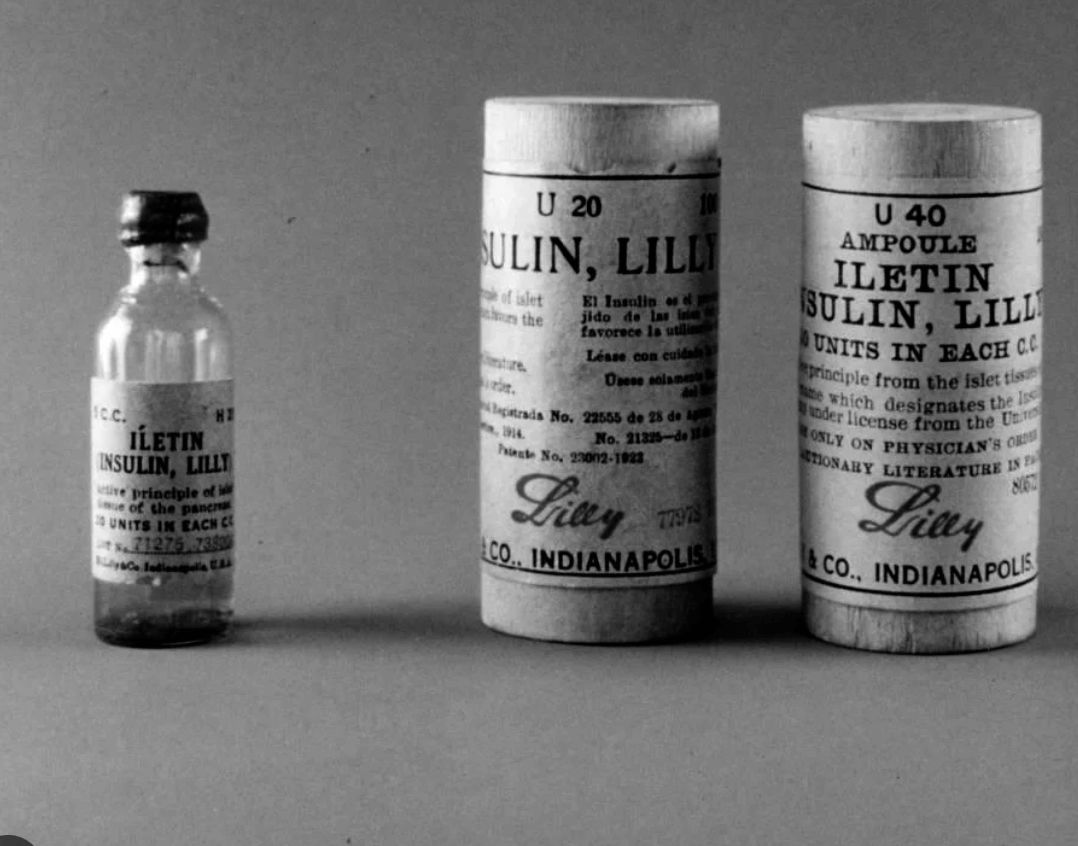
But this ‘miracle drug’ was dependent on extracting the insulin from pigs and cows, using the waste products – pancreases — of the meat packing industry. According to the article in Diabetes Forecast, more than two tons of pig parts were needed to extract just eight ounces of purified insulin.

Over the next half century, the purified insulins obtained from the pig or cow pancreases, which differ slightly in chemical composition from human insulin, were constantly improved in purity and formulated in ways that offered physicians and diabetic patients greater control over blood sugar.
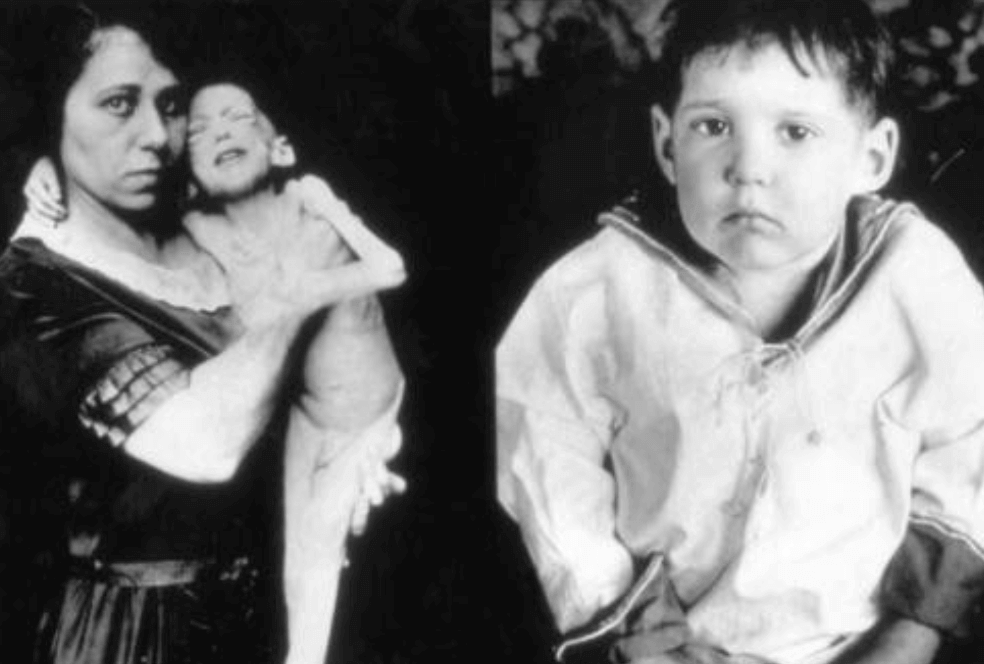
The extracted insulin was, almost literally, a miracle drug. The picture below, dating to the 1920s, shows a patient before (left) and two months after (right) getting insulin.
Early development of genetically-engineered insulin
By the early 1970s, a crisis emerged: As the supply of animal pancreases declined and the prevalence of insulin-requiring diabetes grew, there were widespread fears of possible future shortages of insulin. Fortuitously, around the same time, a new and powerful tool — recombinant DNA technology, also known as “genetic engineering” or “gene-splicing” — became available, offering the promise of unlimited amounts of insulin that, unlike the insulin from animals, was identical to the molecule produced by humans.
The seminal molecular genetic engineering experiment was reported in a 1973 research article by academic scientists Stanley Cohen, Herbert Boyer, and their collaborators. They isolated a ringlet of DNA called a “plasmid” from a bacterium, used certain enzymes to splice a gene from another bacterium into that plasmid, and then introduced the resulting “recombinant,” or chimeric, DNA into E. coli bacteria.

When these now “recombinant” bacteria reproduced, the plasmids containing the foreign DNA were likewise propagated and produced amplified amounts of the functional recombinant DNA. And because DNA contains the genetic code that directs the synthesis of proteins, this new methodology promised the ability to induce genetically modified bacteria (or other cells) to synthesize desired proteins in large amounts.
Lilly immediately saw the promise of this technology to produce unlimited quantities of human insulin in bacteria. After obtaining the recombinant E. coli bacteria that synthesized human insulin from biotech startup Genentech, Inc., they developed processes for the large-scale cultivation of the organism (in huge fermenters like those used to make wine or beer) and for the purification and formulation of the drug.
Insulins had long been Lilly’s flagship products, and the company’s expertise was evident in the purification, laboratory testing and clinical trials of human insulin. The company’s scientists painstakingly verified that their product was extremely pure and identical to pancreatic human insulin (which differs slightly in chemical composition from beef and pork insulin).
Lilly began clinical trials of its human insulin in July 1980. The product performed superbly. There were no systematic problems with treating “naive” patients (who had never received injections of insulin) or those who switched from animal to human insulin. A small number of patients who had had adverse reactions of some kind to the animal insulins tolerated the human insulin well.
The speedy FDA speedy review process
The dossier that provided evidence of safety and efficacy was submitted in May 1982 to the FDA, where I was the medical reviewer and head of the evaluation team. Over many years the FDA had had prodigious experience with insulins and with drugs derived from various microorganisms, so it was decided that no fundamentally new regulatory paradigms were necessary to evaluate the recombinant human insulin.
In other words, recombinant DNA techniques were viewed as an extension, or refinement, of long-used and familiar methods for making drugs. That proved to be an historic, precedent-setting – and correct — decision.
Based on my FDA team’s exhaustive review of Lilly’s data, which were obtained from pre-clinical testing in animals and later in clinical trials involving thousands of diabetics, FDA granted marketing approval for human insulin in October 1982. The review and approval took only five months when the agency’s average approval time for new drugs was 30.5 months.
In retrospect, that rapid approval was particularly remarkable for a drug that was produced with a revolutionary new technology, and that after approval would be available in pharmacies nationwide to millions of American diabetics.
The back story, however, is revealing. My team and I were ready to recommend approval after four months’ review. But when I took the packet to my supervisor, he said, “Four months? No way! If anything goes wrong with this product down the road, people will say we rushed it, and we’ll be toast.”
That’s the bureaucratic mindset. I don’t know how long he would have delayed it, but when he went on vacation a month later, I took the packet to his boss, the division director, and he signed off.

That anecdote is an example of Milton Friedman’s observation that to understand the motivation of an individual or organization, you need to “follow the self-interest.” A large part of regulators’ self-interest lies in staying out of trouble. One way to do that, my supervisor understood, is not to approve in record time a product that might experience unanticipated problems.
The Humulin approval had significant effects. A front-page New York Times article quoted my prediction that the speedy approval was a major step forward in the “scientific and commercial viability” of recombinant DNA technology. “We have now come of age,” I said, and potential investors and entrepreneurs agreed. Seeing that biopharmaceuticals would compete with other medicines on a level playing field, the “biotechnology industry” was on the fast track.
The current burdensome FDA review process
Unfortunately, the rapid approval of human insulin proved to be an anomaly. Even with a toolbox of improved technologies available to both the FDA and industry, bringing a new drug to market on average now takes, on average, 10-12 years and costs, on average, more than $2.5 billion, There hasn’t been much improvement in review time from drug reviews in the pre-electronic era when New Drug Applications were submitted in paper form. (The paper pile for the review of insulin that my group performed was about three times as large as what is pictured in the FDA archive photo at the top of the piece)
Regulators are highly risk-averse, few new drugs are approved now without convening extramural advisory committees, and decisions are sometimes hijacked by political forces outside the FDA. Even so, five of the highest-revenue drugs in the U.S. in 2022 were produced by biotechnology, either with recombinant DNA or monoclonal antibody technology.
Other FDA-regulated biotech sectors have fared much worse. Regulators have made a colossal mess of the regulation of genetically engineered animals, which FDA bizarrely chose to regulate as “new animal drugs” based on their interpretation of a 1938 law. That byzantine process led to a grotesquely prolonged, 20-plus FDA year review of a faster-growing Atlantic salmon.
More disarray occurred with genetically engineered mosquitoes to control mosquitoes that carry viral diseases: Inexplicably, it took FDA more than five years to decide that, for regulatory purposes, this “gene drive” technology was a form of pesticide, and that jurisdiction belonged at the Environmental Protection Agency. As a result of this bureaucratic bungling, the entire biotech sector of genetically engineered animals is moribund.
Government regulation has not aged as gracefully as genetic engineering technology itself, which has advanced significantly over the years. Regulators are supposed to abide by the “bargain” that society has made with them: Civil servants are granted lifetime tenure and are protected from political pressure and retaliation, in return for which they are supposed to make decisions based solely on the public interest. But, often, they do not.
To get FDA-regulated products to those who need them, congressional oversight must emerge from hibernation and create a healthier, more constructive balance.
Henry Miller, a physician and molecular biologist, is the Glenn Swogger Distinguished Fellow at the American Council on Science and Health. He was the founding director of the Office of Biotechnology at the FDA. Find Henry on X at @henryimiller
































