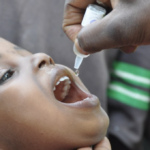
One of the highly anticipated trials of an Ebola drug that showed promising results in monkeys has been stopped early after it apparently failed to show a benefit to patients.
The company that developed the drug, Tekmira Pharmaceuticals of Burnaby, Canada, and the Wellcome Trust, which sponsored the trial, announced that they would not enroll any more patients because the trial had reached “a predefined statistical endpoint.” Early results suggest that adding more patients to the study “was unlikely to demonstrate an overall therapeutic benefit to patients,” the Wellcome Trust said in its statement.
Scientists still have to analyze the data collected to learn more about how well the drug, called TKM-Ebola-Guinea, was tolerated and what specific effects it had on disease outcomes, says Peter Horby of the University of Oxford in the United Kingdom, who headed the study. The trial, which started in March in Port Loko, Sierra Leone, aimed to enroll 100 patients. The company did not say how many patients had been enrolled so far.
The other drug seen as the best shot at fighting Ebola, the antibody cocktail ZMapp, is still being evaluated in a trial. The National Institute of Allergy and Infectious Diseases (NIAID) launched the trial in February in Liberia and the United States. It later expanded to Sierra Leone and is about to enroll patients in Guinea as well, says Anthony Fauci, who heads NIAID, located in Bethesda, Maryland.
The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis. Read full, original post: In setback for potential Ebola drug, company halts trials