The GLP aggregated and excerpted this blog/article to reflect the diversity of news, opinion and analysis.
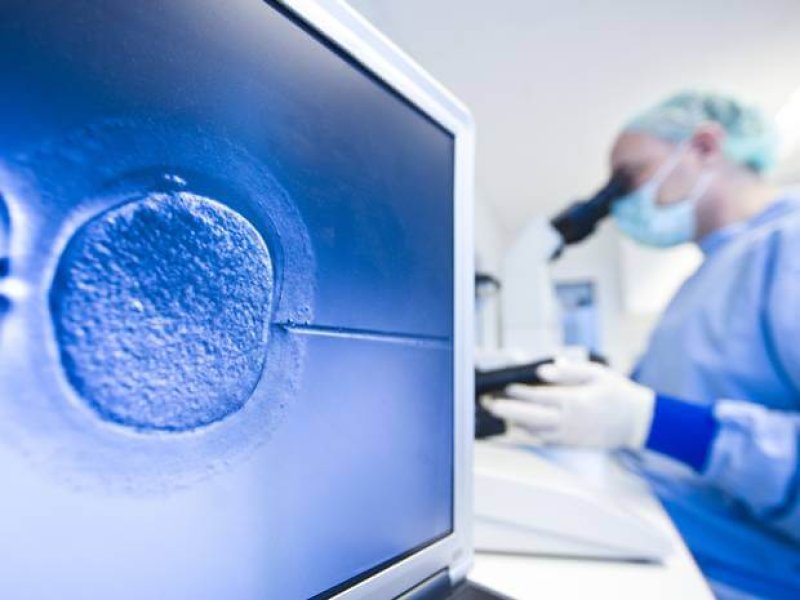
Women having IVF can now incubate embryos in their own bodies before they are implanted in the womb. Results from a clinical trial suggest the incubation device could work as well as conventional IVF and be far cheaper.
Cylindrical in shape, INVOcell is held in the vagina by a flexible diaphragm. The embryos are kept in an inner chamber at body temperature and gases such as carbon dioxide and oxygen diffuse in and out at levels matching natural fertilisation. After five days the embryos will have grown into balls of about 100 cells. The device is then removed and doctors choose the embryo that looks healthiest to implant.
In a U.S. trial of 40 women, the device performed almost as well as conventional incubation, with 65 percent of the women becoming pregnant regardless of the method used. Fifty-five percent who used in-body incubation went on to give birth compared with 60 percent who had the standard method. “We were very pleased with the results,” says Kevin Doody of the Center for Assisted Reproduction in Bedford, Texas, who carried out the trial.
Doody wants to offer the device to women at his clinic, and reckons that it could halve the cost per cycle, typically $16,000 to $20,000 in the U.S. In conventional IVF, incubators are set up to mimic the body and have to be regularly monitored to ensure early embryos are supplied with the right amounts of gas and that conditions are optimal for five days. Because the woman’s body acts as a natural incubator, such expensive equipment isn’t needed, he argues.
Read full, original post: Women can ‘grow’ their own IVF embryos with in‐body incubator