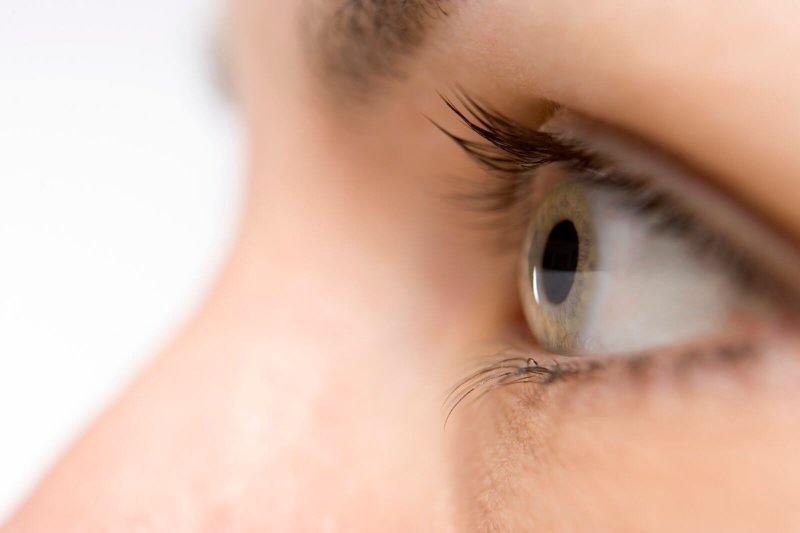
The $850,000 list price for a new medicine that treats a genetic form of childhood blindness is about four times too high for the value the drug provides, a nonprofit that studies the cost-effectiveness of new drugs said [on January 12].
…
The report from the Institute for Clinical and Economic Review focused on the medicine Luxturna, the first-of-its-kind gene therapy approved for the U.S. market and the most expensive medicine by list price. It is the latest flashpoint in the debate over how to afford an innovative medicine — in this case, a therapy that corrects a genetic mutation in people’s cells — that carries, and in some views, deserves, a pricey list cost.
In its report, ICER said a cost-effective price for Luxturna would be $153,000 to $217,000 — a discount of 75 percent or more. ICER cited a lack of data that Luxturna causes permanent improvements in vision as a key reason that its developer, Spark Therapeutics, should not be charging so much.
…
While Luxturna is a one-time therapy, it does not “cure” people born with mutated RPE65 genes; instead, it is meant to stop disease progression and restore some visual strength. But other gene therapies in pipelines could, by replacing faulty genes with functional ones, actually amount to cures.
Read full, original post: At $850,000, price for new childhood blindness gene therapy four times too high, analysis says