On June 30, some 80 environmental organizations, led by Greenpeace EU, Friends of the Earth Europe and Save Our Seeds, signed an open letter to the European Commission asking for support for a global moratorium on gene drive technology. The advocacy groups claimed that the release of gene drives “poses serious and novel threats to biodiversity and the environment at an unprecedented scale and depth”.
Citing a report by the European Network of Scientists for Social and Environmental Responsibility (ENSSER), the coalition wrote:
…in light of the unpredictabilities, the lack of knowledge and the potentially severe negative impacts on biodiversity and ecosystems, any releases (including experimental) of Gene Drive Organisms into the environment be placed on hold to allow proper investigation until there is sufficient knowledge and understanding.
The environmental claims were unsupported by any documents other than the “report” by ENSSER, a controversial group of anti-biotechnology activist scientists co-founded by Gilles-Éric Séralini, best known for his retracted and discredited 2012 paper linking GMOs to cancer in rats.
The European parliament has already supported such a moratorium, an act that echoes EU’s precautionary approach to genetic engineering, transgenic organisms and gene editing. The EU stated reasons include:
- Human-induced extinction of species
- Irreversible changes to food chains and ecosystems
- Loss of biodiversity
- Mutations could happen mid-drive, which has the potential to allow unwanted traits to unexpectedly “ride along”
- Cross-breeding or gene flow potentially allow a drive to “escape” and move beyond its target population
- Unknown ecological side effects Ecological impacts: Even when new traits’ direct impact on a target is understood, the drive may have side effects on the surroundings
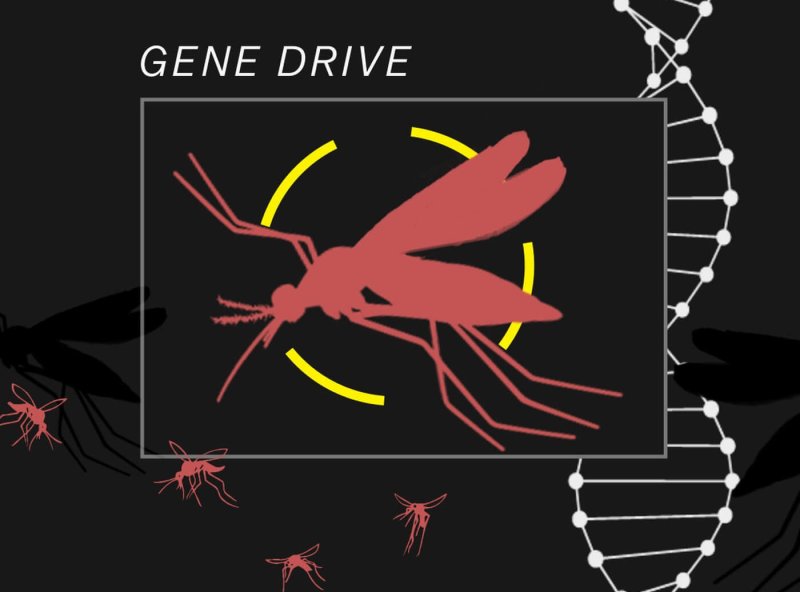
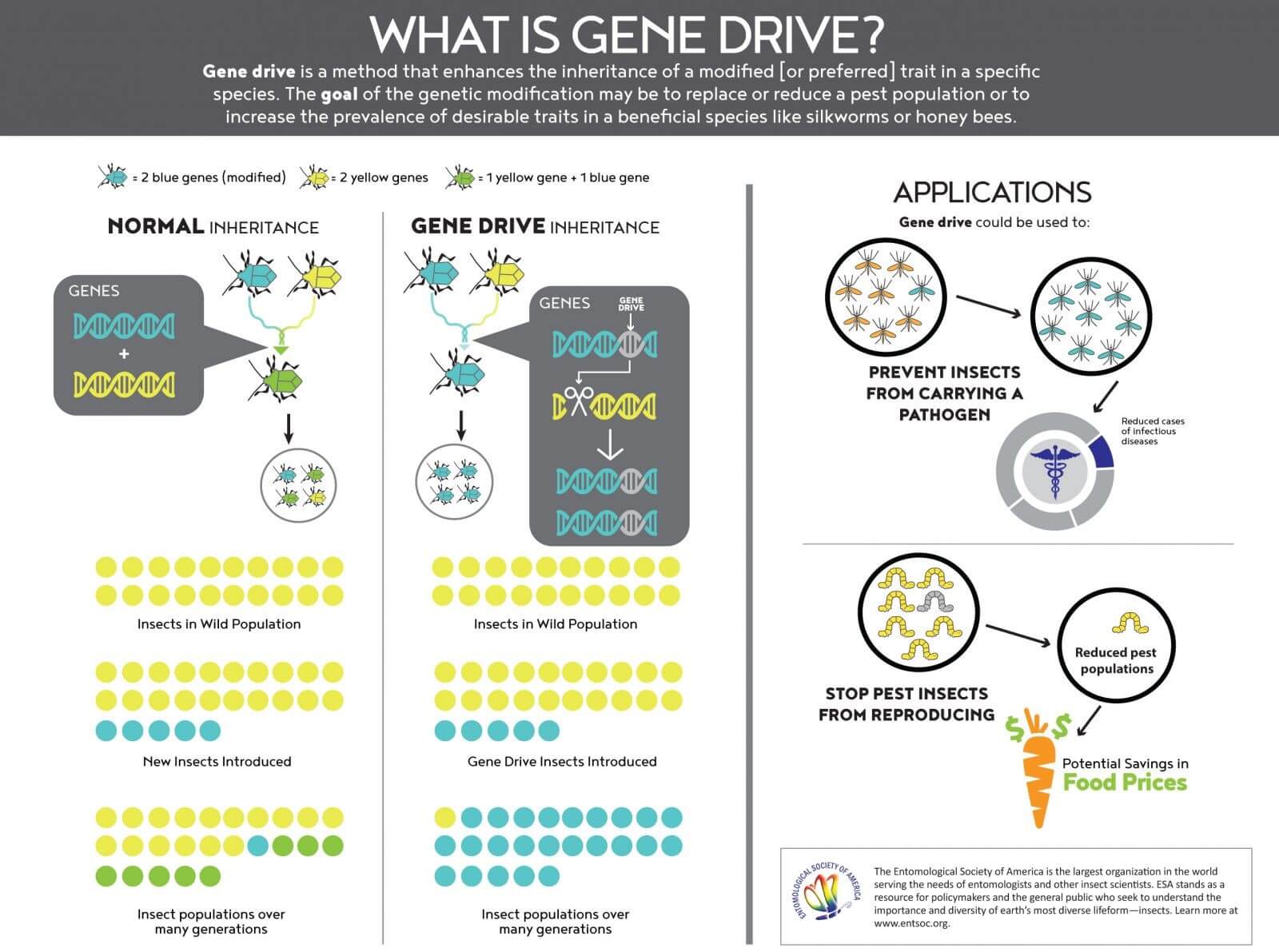
What are gene drives?
Recent advances in genetics and synthetic biology, particularly the development of CRISPR gene editing tools, have given scientists a powerful way to address problems created by pests, from mosquitoes to rodents, that vector disease to humans. In classical genetics, genes that offer adaptation benefits to individuals tend to increase their occurrence in the population while genes that reduce fitness tend to disappear.
Gene drives are genetic sequences designed to spread strongly and become present in every individual of a targeted species after a few generations. The genes may offer benefits, be neutral for adaptation purposes, or hinder their carriers’ survival and reproduction potential.Generation after generation, it would relentlessly copy and paste the gene it carried, until the gene and the desired trait was present in every descendant. Because the spread of a trait happens over generations, a gene drive works best in species that reproduce quickly, like insects and rodents
Gene drives are the first genetic constructs that can theoretically affect a population in its entirety, and quickly. It could even lead to the extinction of entire species, as gene drive critics allege. Species extinction has been part of life and evolution for all of Earth’s history. Although the data are fuzzy and contested, the UN Convention on Biological Diversity concluded that 150-200 plant, insect bird, and mammal species go extinct every day.
The likelihood that a gene drive will destroy a species in part or in whole, such as the infected Aedes aegypti mosquito species that carries the Zika, dengue and chingunya viruses and offers no known environmental benefits, is nonetheless daunting to some. On the one hand, gene drives could be used to eradicate disease such as malaria and yellow fever by controlling the mosquitoes that transmit them. On the other hand, critics fear that the technology will open a Pandora’s Box; removing a species that theoretically could resultin what is popularly and controversially known as the “butterfly effect”.
As imagined by MIT meteorologist Edward Lorenz 60 years ago, a tiny environmental change—say an extinction of a pest—could dramatically and unpredictably result in unpredictable or even catastrophic consequences (Lorenz imagined a butterfly flapping its wings and causing a typhoon).
In the last few years, various groups have called for a global moratorium on gene drives. Such attempts were resisted at the 2016 and 2018 United Nations Conventions on Biological Diversity, mainly due to the strong opposition of many scientists and sub-Saharan African nations hardest hit by disease-vectored pests. Nevertheless, gene drive opponents have gained traction and gene drive research and applications face significant regulatory obstacles across the world (see Genetic Literacy’s Global Gene Editing Regulation tracker for a country-by-country analysis).
What does the scientific evidence say about gene drives and their environmental consequences?
Myth 1: The eradication of a population will destroy the ecosystem
There are over 3,000 mosquito species, likely a fraction of the number of species that have existed over some 100 million years. A handful of these (Aedes, Anopheles, and Culex species) are disease vectors and transmit infections such as malaria, yellow fever, the West Nile virus, Zika, and dengue fever. Mosquito-borne disease account for more than 17% of all infectious diseases and cause more than 700,000 deaths every year. These mosquitoes are mostly invasive in their ecological distributions.
“Ultimately, there seem to be few things that mosquitoes do that other organisms can’t do just as well—except perhaps for one,” reported Nature magazine in a 2010 article “A World Without Mosquitoes.
“They are lethally efficient at sucking blood from one individual and mainlining it into another, providing an ideal route for the spread of pathogenic microbes.” The Nature article concluded that wiping out mosquitoes wouldn’t be a bad thing. In fact, they could restore rather than harm the ecosystem. The same can be inferred for most parasitic insects, which are specialized to a particular host and normally don’t have an extended ecological interactions network.

Invasive species also cause significant environmental hazards. Cane toads, having no natural predators, are slowly taking over the Australian continent from the northeast. Invasive fish from the red sea are wrecking havoc in the Mediterranean marine ecosystems. Rodents have spread in every conceivable corner of the earth, displacing vulnerable local fauna.
Gene drives might be one of the only ways to contain their spread, protecting biodiversity. They can be a powerful conservation tool that targets only the organism of interest, unlike contemporary pest management techniques such as the use of insecticides that attack all insects indiscriminately, or introduction of natural predators from other ecosystems (that by default disturb the food chains and interactions network).
Myth 2: Gene drives can jump from species to species and spread uncontrollably
It is possible for a DNA sequence to jump from one species to the other through a process called horizontal gene transfer. This theoretically could happen between insects, which appears to lend support to the argument that there is at least a small chance for a gene drive to move from species to species with unforeseen consequences.
The truth is that gene drives can be designed to target a very specific area of the genome, unique for a species. The modern gene drives use the precise CRISPR base editing technologies to spread to the population. In the off chance that the DNA encoding the gene drive will enter the reproductive cells of an individual from the other species, the editing system will have no template to act upon and the gene will be lost. One may argue that CRISPR has a chance for off-target activity, but a gene drive needs maximum efficiency to act as a gene drive. If the CRISPR doesn’t work at 100%, the DNA sequence will be subject to the typical laws of inheritance and will disappear from the genetic pool
Valid concern 1: Gene drives could be used by the military
The ability to introduce genetic information to a wild population, which will spread to every individual, is unfortunately a dual use technology. The technology can theoretically be exploited to make biological weapons, though there’s no indication that such a weapon is or has been developed. As gene drives can work well across many generations and require a large amount of offspring, they are unable to directly harm humans, crops, and farm animals. But a gene drive could be used to enhance the fitness of a crop-eating insect or a disease-carrying rodent.
The solution to this potential hazard is more research (and definitely not a research moratorium). Anyone with the means (which are considerable, so no lone bioterrorists or rogue scientists) and intent to cause harm can already research into such applications and will ignore aUN-imposed technology ban. The research community needs to develop the means to detect and monitor any malicious gene drive release and counter any offensive use.
Valid concern 2: Who decides and who is responsible?
The question on who and how should approve gene drive projects isn’t easy to answer. A gene drive isn’t contained by country borders, and the outdated GMO regulation framework existing in most countries is scientifically outdated and practically inadequate to handle such applications.
Moreover, the technology cannot be monopolized by a few countries or private companies. Each project is different. The approval should be a result of consensus among numerous stakeholders. There should also be a defined way to monitor how the gene drive spreads and how to handle liability claims if there are negative effects.
With populism growing and fewer people willing to trust the judgment of regulators and scientists, the rhetoric around complex innovations has become increasingly polarized, with both sides stuck fighting a high-stakes battle for public opinion. The issue is complex, and any decisions cannot be left to scientists, state organizations, and companies alone. But it also cannot be left solely in the hands of environmental organizations with little or no understanding of the science and with an ideological agenda that doesn’t necessarily serve the public.

Environmental groups have often resorted to hyperbole as the debate over gene drives has unfolded. At the UN Convention on Biological Diversity in Sharm el Sheikh, Egypt, in 2018, a coalition of activists compared gene drives to the atomic bomb and accused researchers of using malaria as a Trojan horse to cover up the development of agricultural gene drives for corporate profit.A handful of small NGOs in the US, collectively known as SynBioWatch, have taken to describing gene-drive researchers as a cabal. The Canadian anti-biotechnology organization ETC Group claims aggressively spreads misinformation on social media, including claims that gene-drive honeybees could supposedly be controlled with a beam of light.

Meanwhile, Florida Keys is experiencing the largest dengue fever outbreak in a decade, with close to 40 cases already documented. The outbreak has led the Florida Keys Mosquito Control District to enter a partnership with UK-based, US-owned Oxitec that could lead to the Keys becoming the first U.S. trial site for genetically modified Aedes aegypti mosquitoes.
With a technology that can prevent hundreds of thousands of deaths per year, it is unethical to peremptorily ban it because it doesn’t fit a few people’s worldview of what is ‘natural’. One may argue that governments and regulators should have no say whether one species should go extinct or not. But one can also question why activist groups in North America or Europe should be able to insert themselves in life and death decisions, preventing initiatives across the globe that could save millions of lives and protect our population’s health and crops, and promote biological diversity.
Kostas Vavitsas, PhD, is a Senior Research Associate at the University of Athens, Greece. He is also a steering committee member of EUSynBioS. Follow him on Twitter @konvavitsas