The US Food and Drug Administration relaxed certain federal regulations [December 16] on medication abortion, a two-medication process that has become one of the most commonly used abortion methods in the United States.
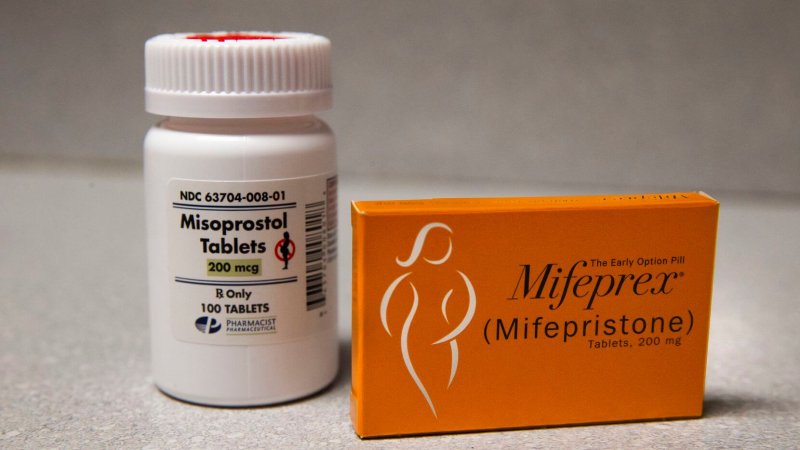
Medication abortion, also known as medical abortion, is a method by which someone ends their pregnancy by taking two pills, rather than having a surgical procedure.
The first pill is mifepristone, sold under the brand names Mifeprex or Korlym. It’s also known as RU 486. The second pill is called misoprostol.
In April, the FDA allowed medication abortion pills to be sent by mail during the Covid-19 pandemic and said it would no longer enforce a rule that required women to get the first of the two pills in person at a clinic or hospital.
The American College of Obstetricians and Gynecologists (ACOG) and several other medical associations have long advocated for the removal of restrictions on how the drugs are administered.
The restrictions, the group argues, “do not make the care safer, are not based on medical evidence or need, and create barriers to clinician and patient access to medication abortion.”