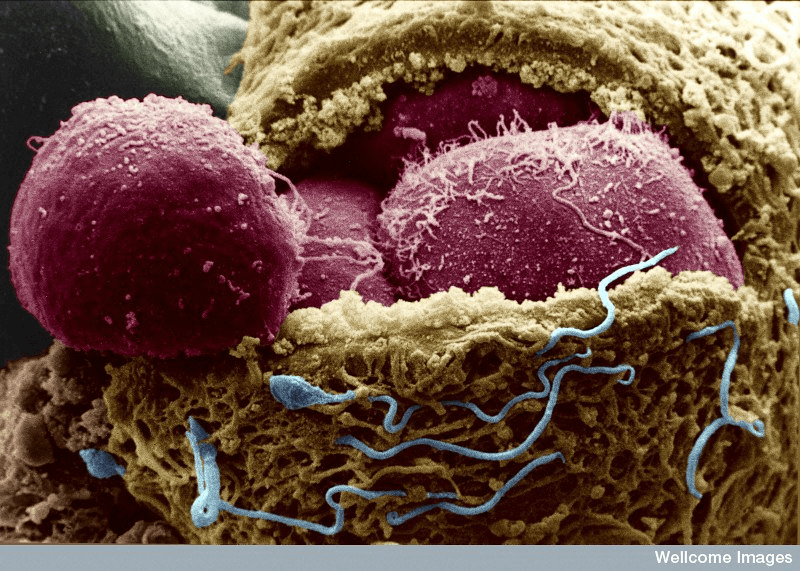
Starting about a decade ago, scientists in a handful of developmental biology labs around the world began cataloging the complex chemical recipe an embryo uses to make gametes — sperm or egg cells — with the idea that if they could copy it, they could coax any cell along the same path. Working mostly in rodents, these pioneers in so-called in vitro gametogenesis, or IVG, have since turned mice stem cells into sperm and eggs, including a recent success creating eggs from the stem cells of a male mouse.
Key to these breakthroughs is first creating a clump of reproductive tissue to send these would-be-gametes the coordinated signals they need to become sperm or eggs. [Researcher Christian] Kramme spent most of his PhD figuring out how to make the human version of this ovary-in-a-dish, supported by New York City-based biotech Gameto.
…
These mini-ovaries, or “ovarioids,” aren’t yet sufficiently like the real thing to produce the worlds’ first lab-grown human egg, a milestone that would open the door to an array of radical new reproductive possibilities. But Gameto licensed the technology for what it sees as a much more near-term (and slightly less sci-fi) application: boosting the odds of success for people seeking in vitro fertilization.