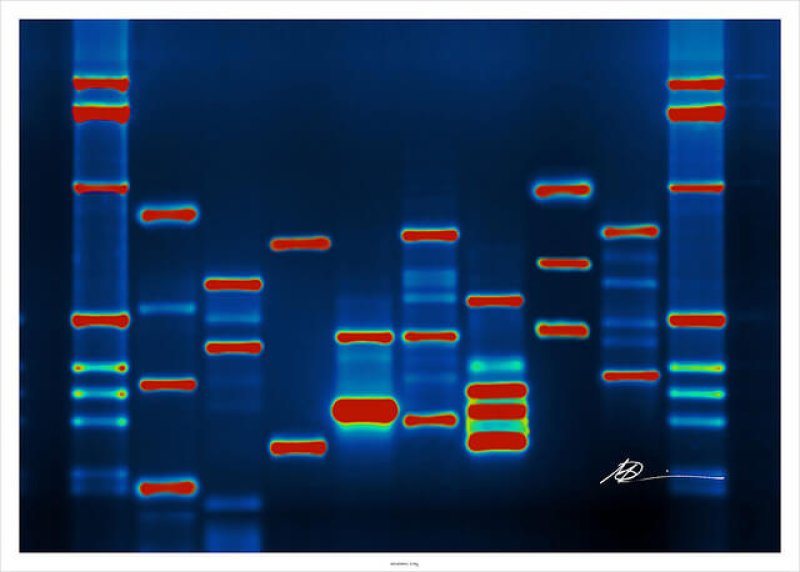
Sequencing, sequencing, sequencing. You’d be forgiven for thinking that the sum total of cancer genomics — indeed, disease genomics of any kind — is gathering more and more and more genome sequences from afflicted individuals. To some extent, that’s what it has been. This is poised to change, however.
A recent Nature editorial outlines some of the major cancer sequencing efforts:
Seventeen countries have invested in sequencing cancer genomes through the International Cancer Genome Consortium (ICGC), which aims to sequence more than 25,000 samples. The largest and oldest component of that project is The Cancer Genome Atlas (TCGA) at the US National Cancer Institute (NCI) in Bethesda, Maryland, which intends to characterize 10,000 tumours.
Its projects like these that have brought us to the point where the UK’s National Health Service (NHS) is planning to work with cancer researchers and pharmaceutical companies with the aim of developing drugs tailored to the genetic profiles of specific patients’ tumors. They’ll start with lung cancer, but according to a report in The Telegraph, and will be treating most cancers this way in five to 10 years.
Even with this milestone approaching, the big sequencing projects mentioned by Nature are winding down. The TCGA and ICGC are no longer seeking new samples and are focused on completing analyses of the samples they do have.
As they complete their analyses, we’ll have a tremendous amount of cancer sequences available (though no where near enough for comprehensive catalog of all cancers). Mission at least partially accomplished. But scientists keep pushing for more sequences?
The Nature editors argue that it’s time to take a new approach. Time to start tracking the trajectory of individual patient’s cancers and mapping that to a sequence of genetic samples. Taking just one sequence of a tumor and adding to a massive, mostly faceless database is no longer going to cut it — we need to learn how cancers change over time. The editors write:
The end of TCGA also represents an opportunity for the field to balance its cancer-genomics projects more evenly between cataloguing mutations and studying their functional significance. Functional studies have had short shrift, whereas sequencing — a simple concept, and easier to communicate to policy-makers and the public — has taken the lead.
The Nature editors only touch upon the biggest bugaboo for the genetics-based treatment of cancers, which is the difficulty we continue to have translating all of this genetic data into clinically useful information. GLP’s Tabitha M. Powledge addressed this in depth on the topic of breast cancer in her feature story from last week. Even dealing with the best-known mutations related to breast cancer — those in the BRCA genes made famous by Angelina Jolie and Myriad Genetics — the variance in risk is huge.
Powledge writes, “Even with the BRCA mutations known to cause disease, the range of risk is […] 40 percent in some people, nearly 90 percent in others, and usually it is not clear which person with the mutation is at highest risk.” And BRCA mutations are far from the only source of breast cancer. “The BRCA genes are the best known, but are involved in only 1 out of 4 cases of breast cancer with a hereditary component. Most breast cancers are not familial. About 70 percent have been considered sporadic.”
This uncertainty plagues all of genetic medicine. A Bio-IT World piece by Aaron Krol emphasizes the confusion that too-often clouds attempts to apply genetics in the clinic. Say someone undergoes a round of genetic testing, after which …
Each variant found in the course of testing – and in a mid-size gene panel there will be hundreds – has to be individually interpreted, taking into account factors like its population frequency, similarity to other variants known to be pathogenic, location within the gene, and, if samples from a patient’s family members are available, whether it segregates with disease.
This level of interpretation naturally leads to discrepancies among labs and clinicians — one might deem a given mutation benign while another would call it “uncertain”. Furthermore, labs end up hoarding data for a variety of reasons; some are too busy to upload their data; some don’t even think to share data that isn’t impressive enough to merit proper publication; and others (Myriad comes to mind) seem locked in a competitive mindset.
The ideal solution, as proposed in Krol’s piece, is a collection well-curated medical genome databases. A mix of radical openness and rigorous oversight to ensure that data are both widely available and watched over for accuracy. Krol is talking about genetic diagnostics generally, but his arguments apply perfectly to cancer genomics.
The advent of genetically tailored drugs and and success of big projects like the TCGA and ICGC are certainly cause for celebration, but they’re also cause for reflection. It’s time for the next phase of cancer genomics, one defined by an emphasis on more clinical data-gathering and less cataloging; more collaboration and less competition.
Kenrick Vezina is Gene-ius Editor for the Genetic Literacy Project and a freelance science writer, educator, and amateur naturalist based in the Greater Boston area.