Research was initiated in the early 1990s which led in 2000 to the publication of the technology behind what came to be known as Golden Rice. From the outset, the intention was to create a source of vitamin A in the endosperm of rice, as an additional intervention for vitamin A deficiency. Philanthropy and the public sector funded the research. In 2001, the inventors, Professor Ingo Potrykus and Dr. (now Professor) Peter Beyer, assigned their patents to Syngenta for commercial exploitation as part of a transaction which obliged the company to assist the inventors’ humanitarian and altruistic objectives.
At the same time, the nutritional technology was donated by its inventors for use in developing countries. The inventors licensed a network of Asian government-owned rice research institutes to deliver their objectives. Product development was initiated through the International Rice Research Institute (IRRI) and the network. The whole network, including IRRI, worked to a common set of goals defined in licences each institution signed with the inventors. The terms included that there would be no charge for the nutritional technology and it would only be introduced to publicly owned rice varieties.
This article is part one of a four-part series on golden rice based on a book chapter Golden Rice: To Combat Vitamin A Deficiency for Public Health. Part 2: Will nutritionally enhanced rice work and help solve malnutrition in developing countries?; Golden Rice, Part 3: A thoroughly studied, safe and nutritionally-enhanced GMO crop approved by Australia, Canada, New Zealand and the US, but vilified by Greenpeace and ‘environmental’ activists; Part 4: Do biofortified crops make economic sense?
Improvements were made to the technology by Syngenta scientists. In 2005 and 2006, pursuant to Syngenta’s legal obligations entered into with the inventors in 2001, Syngenta provided selected transformation events of the improvements to the Golden Rice Humanitarian Board. The Humanitarian Board, via Syngenta and IRRI, made these new versions available to the Golden Rice licensee network. In 2004 Syngenta ceased its commercial interest in Golden Rice. From 2004 development was again only funded by philanthropy and the public sector; the national budgets of Bangladesh, China, India, Indonesia, Philippines and Vietnam; as well as the US National Institutes of Health together with the Rockefeller and Bill & Melinda Gates Foundations and USAID. Golden Rice is a not-for-profit project: no individual, nor organization involved with its development, has any financial interest in the outcome.

To date the Golden Rice project has principally engaged plant scientists. Activist opposition to Golden Rice has been led principally by non-scientists, who have been very successful in developing a narrative about Golden Rice and GMO crops which serves the activist’s purpose but is fundamentally inaccurate. Further background to the development of Golden Rice, including the political dimensions, is detailed elsewhere.
A few years ago, at Tufts University, USA, I gave a presentation about Golden Rice. The symposium was organized by the Friedman School of Nutrition Science and Policy whose strategic aims today include ‘Reduce nutrition-related health inequities’ and ‘Promote food systems that increase agricultural sustainability while improving human health.’ I was dismayed to learn that the anti-GMO and anti-Golden Rice activists’ narrative was widely accepted by the participants—all of whom were studying or working in nutrition and well aware of nutritional inequities in public health.
Without adoption, that is, regular growth and consumption of Golden Rice by populations in countries where rice is the staple and VAD is problematic, Golden Rice cannot deliver any public health and welfare benefits. Adoption requires cooperative working by different specialists, including medical, nutritional and public health specialists. This chapter is designed to answer anticipated questions from such specialists, to facilitate adoption of Golden Rice as an additional intervention for vitamin A deficiency.
Rice, diet and deficiency
Rice is the most important staple crop: more than half of the global population eats it every day. In some countries, 70–80% of an individual’s calorie intake is from consumption of rice.
For storage without becoming rancid, the husk and the aleurone layer of rice have to be removed. What remains after polishing–white rice, the endosperm–contains small amounts of fat and is an excellent source of carbohydrate for energy but contains no micronutrients. Yet humans require both macronutrients (carbohydrates, proteins, fats) and micronutrients (minerals and vitamins) for a healthy life. Like all plants, rice obtains its minerals from the soil. Vitamins are synthesized by plants and/or animals, including humans.
Human health is best served by a ‘balanced diet’ that is varied, containing both macronutrients and micronutrients, including animal products and, as sources of provitamin A, coloured fruits and vegetables. Micronutrient sources are insufficiently represented in the diets of many people in countries where rice is the staple. The reasons often include poverty: such dietary components are expensive compared to the cost of rice. In countries where rice is the staple, the average consumption is 75.20 kg/capita/year. Of those countries where micronutrient deficiencies are common, consumption increases to 150 kg/capita/year. In such populations micronutrient deficiencies, like poverty itself, often occur as part of an intergenerational cycle.
For the past 15 years, 800 million people—more than 10% of the global population—are hungry every day. These chronically hungry individuals lack sufficient calories in their daily diet; indeed over the past 3 years, the trend is upward. Even more alarming is that 2 billion people—almost 25% of global population—are micronutrient deficient; they suffer from ‘hidden hunger,’ with important associated morbidity and mortality and related economic impact. Figure 1 shows that over the 20-year period 1990–2010, the rate of reduction of chronic hunger (that is, macronutrient—carbohydrate, proteins and fats—dietary insufficiency) has been faster than the rate of reduction for hidden hunger (that is, dietary insufficiency of minerals and vitamins).
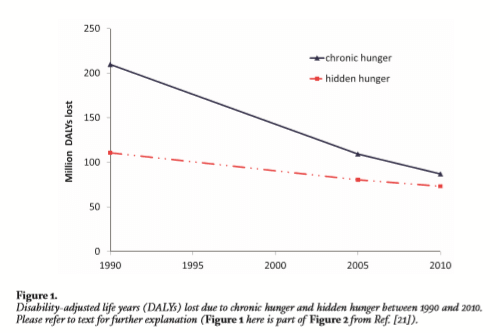
Dr. Matin Qaim, member of the Golden Rice Humanitarian Board and one of the authors of the paper from which Figure 1 is extracted, has commented: ‘In the future the hidden hunger [e.g. micronutrient deficiency] burden will be larger, [than chronic hunger – principally carbohydrate deficiency] unless targeted efforts to reduce micronutrient malnutrition are implemented at larger scale.’
Interventions for micronutrient deficiencies include supplementation (with pills, syrups or capsules containing micronutrients) and fortification (adding micronutrients to processed food). Both interventions require some level of manufacturing and/or distribution infrastructure.
With the creation of Golden Rice in 1999—the first purposefully created biofortified crop—a new term was required: ‘biofortification.’ The word was first used in 2002 and first defined in 2004: “biofortification” is a word coined to refer to increasing the bioavailable micronutrient content of food crops through genetic selection via plant breeding.’ In 2003 ‘Harvest Plus’ a not-for-profit public-sector programme started to biofortify staple crops by conventional plant breeding, to benefit the poor, and progress with biofortification through conventional plant breeding was rewarded by the World Food Prize in 2016.

The intention of biofortification is to deliver public health benefits to populations which are micronutrient deficient, through consumption of the staple crop including the extra nutrition within the edible part of the crop. In this way minimal cultural change is required to food—production, processing or consumption—systems. For the most marginal members of the population, this biofortification approach overcomes the inherent access, cost and non-sustainability difficulties of supplementation and fortification. In 2017 the World Bank recommended that biofortified staple crops should be the norm rather than the exception: ‘conventionally’ bred biofortified crops and also genetically engineered crops—gmo crops—were both recommended with Golden Rice specifically mentioned.
For Golden Rice to deliver benefits, it has to be grown and consumed within target countries where VAD remains problematic despite significant progress with other interventions, notably vitamin A capsules, which have undoubtedly saved millions of lives and will save more, since they were introduced (accompanied by controversy) in the 1990s. And success or failure with Golden Rice will directly affect future adoption also of high zinc, high iron and high folate rice and their impact on public health for hundreds of millions of people. All these traits, introduced to the endosperm of rice, necessitated using GMO techniques, and all cost no more than white rice to the grower or consumer. Eventually, as the end point of product development, it is planned to include all these nutritional traits together in multi-micronutrient-Golden Rice.
Adoption of Golden Rice requires public health professionals as well as agricultural and other professionals, to work together in each country. Any skepticism created by the past 18 years of negative activist influence will prevent success, if not positively addressed by all involved. For billions of people, the stakes could not be higher.
Adrian Dubock holds a PhD from Reading University in reproductive physiology and ecology. He is the executive secretary of the Golden Rice Humanitarian Board































