Mosquitoes are not just obnoxious summer pests — they are a serious health threat to most of the world. In fact, the WHO calls mosquitoes “one of the deadliest animals in the world.”
Why? Mosquitoes carry and spread diseases to humans that cause millions of deaths every year. The biggest threat is malaria: a half a million lives are lost annually, and Africa alone loses $12 billion in health care, productivity, investment, and tourism to the disease. Then there’s Zika, dengue, chikungunya, and yellow fever — each carried by mosquitoes, and each extracting their toll in human lives and livelihood.
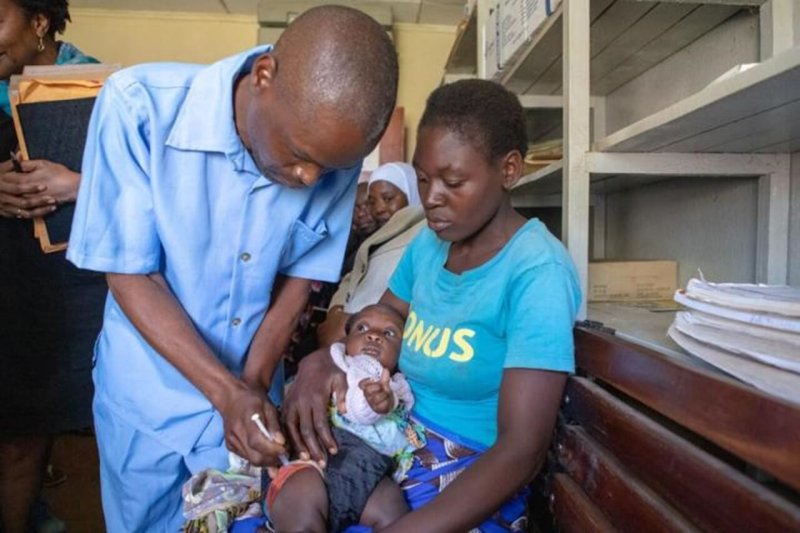
But now, there is hope that biotechnology can help solve this crisis. The solution lies in genetically modifying a small population of mosquitoes and releasing them into malaria-prone areas. These mosquitoes carry a lethal gene that kills larvae before they reach adulthood and carry malaria to others, just stunting human illness.
Oxitec is a British biotechnology company doing just this. Over the last fifteen years or so the company has introduced Friendly™ Technology. Oxitec’s latest results back up historic successes in Brazil, which saw dengue cases in one area reduced by 91% in a small trial. Their latest study showed a 96% reduction in mosquito numbers, this time using a more effective strategy of targeting the biting, egg-laying females – albeit leaving non-biting males to survive and reproduce. A great success, on the face of it.

The anti-GM backlash
However, Oxitec and others are taking a lot of flak from the anti-GM lobby, which according to the Genetic Literacy Project spent $850 million in the last five years alone opposing everything from the way we label genetically engineered food to fabric that’s fermented from sugar.
The backlash was triggered by a paper that, ironically, seems likely to be retracted (or at least highly modified) due to exaggerated predictions of more robust mosquitoes, among others. Though reports of genetic mixing between surviving introduced mosquitoes and local mosquitoes were valid, the lethal gene was not actually present – which was the most likely outcome considering the lethality of the gene in question. A lethal gene will naturally diminish in frequency among a population.
Those 219 million annual malaria cases cost low income countries a significant chunk of their GDP — up to 1.3% in the worst affected — while just a 10% reduction in malaria was associated with a 0.3% growth in a much cited study on the economic burden of malaria. For Uganda, the economic gain from eradicating malaria would total around $50 million USD.
This doesn’t take into account emerging diseases such as dengue and Zika virus, among others. In Brazil, the focus of Oxitec’s recent trials aiming to reduce numbers of Aedes aegypti, 1.2 million people were infected with dengue in the first six months of 2019 — six times more than in 2018 — with an associated 388 deaths. Zika virus, carried by the same mosquito, exploded onto the scene in 2015 and has been associated with a range of effects on babies born to infected mothers in 10% of cases.
Yes, there are likely some knock-on ecosystem effects of releasing Oxitec mosquitoes. There might well be reduced prey for fish that eat the larvae, or less food for some of the birds that eat the flying insects. However, mosquitoes are not irreplaceable as far as the wider ecosystem is concerned, especially pests such as Aedes aegypti. There are another 3000 species of mosquito other than the three which primarily cause disease in people.
The World Health Organization (WHO) brands Aedes aegypti mosquitoes as being “exquisitely adapted to city life”, and that they “prefer to breed in artificial containers”. Given this information, it makes it even less important that lethal genes might persist among the wider population of these costly pests, less so that they might be eradicated – an opinion seemingly shared by researchers who have looked into the potential environmental costs associated with getting rid of the worst species of disease-bearing mosquitoes.
It all boils down to a cost/benefit analysis. Is the risk of some genetically modified mosquitoes passing on transgenes to wild populations worse than the risk of millions of people being infected with haemorrhagic fever and malaria, or babies being born with abnormally small heads (microcephaly)?
In any case, we will always have to take measures to reduce the burden of disease. What about the alternative forms of pest control and their relatively indiscriminate, off-target effects?
What is natural?
Oxitec, the company responsible for releasing genetically modified mosquitoes, have always known and stated – explicitly so – that some mosquitoes would survive to breed and pass on their genes. They have also taken measures to ensure that populations containing the lethal gene eventually go their predestined way.
In fact, their latest mosquitoes are meant to pass on genes to wild populations, this time to reverse the naturally occurring genetic mechanisms that render mosquitoes resistant to pesticides such as DEET. (Incidentally, multi drug resistance in the malaria parasite itself is also increasing).
To put our collective minds at ease, it’s worth pointing out that nature is weirder than what Oxitec is doing. mosquitoes perform their own version of enforced sterilisation, whereby male tiger mosquitoes (of the species Aedes albopictus) can mate with, and sterilize, female Aedes aegypti mosquitoes.
The fact that diseases such as Zika and dengue are present in Brazil in the first place is evidence that we are about as far removed from whatever “natural” used to mean as we can possibly be. Zika was introduced by a traveller from French Polynesia. The only way it got to Brazil was by aeroplane.
It’s not just Zika traversing the globe in such a manner. There are emergent diseases popping up left, right and center, and it is not an easy task to keep them in check. The WHO warned us over ten years ago that infectious diseases are emerging at a rate not seen before. (One example is West Nile Virus, a mosquito-borne disease first described in 1937 in Uganda but discovered in New York in the summer of 1999. It is becoming more prevalent in California and will soon reach Silicon Valley and the San Francisco biotech region.)
It is no wonder. We live in a globally connected world with a rapidly expanding population that is a hotbed for disease-causing agents to emerge, mix, and spread (from viruses and bacteria to malaria parasites), which means that we need highly innovative, modern solutions to control them. It’s an evolutionary arms race, and we need all the tools we can muster. If one of them happens to include lethal genes that successfully wipe out local populations of disease-causing mosquitoes, so be it.
Mosquito-borne diseases already threaten half of the global population.And as the climate warms and favors the mosquito, these diseases could spread to a billion more people.
Clearly, our current methods of control haven’t quite been enough to stop 219 million people becoming afflicted with malaria each year, or an increasing number suffering and dying from dengue and other diseases. It’s not genes escaping that is the problem, but the sheer difficulty in eradicating mosquitoes and their diseases at all.
We must proceed carefully with new genetic engineering technologies, but we must also weigh the risks of inaction: each year hundreds of millions of people — mostly children — needlessly die, get sick, or suffer genetic defects. GMO mosquitoes are something we can do about it.
So what would you rather have: GMO mosquitoes or dying babies?
John Cumbers is the founder and CEO of SynBioBeta, the leading community of innovators, investors, engineers, and thinkers who share a passion for using synthetic biology to build a better, more sustainable universe. He is an operating partner and investor at the hard tech investment fund Data Collective, and a former bioengineer at NASA. Follow him on Twitter @johncumbers and @SynBioBeta
A version of this article was originally published on Forbes’ website as “What’s Worse: GMO Mosquitoes Or Dying Babies?” and has been republished here with permission.