Many tests take days to produce results, require leaving quarantine to visit a medical professional, or — most likely — both.
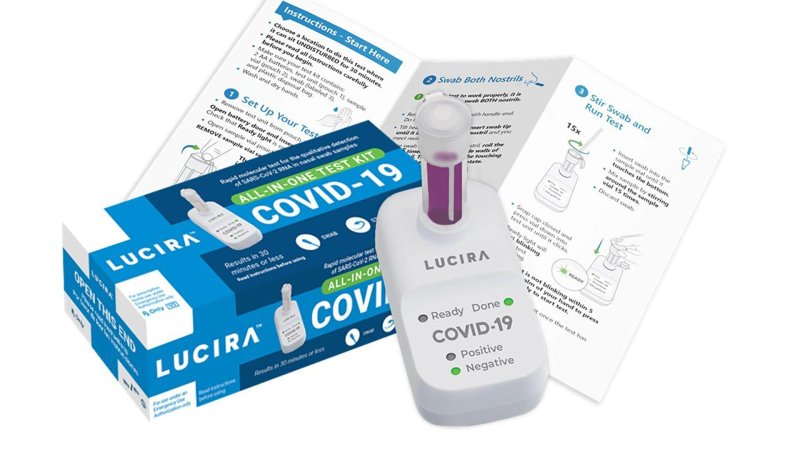
That could change with Lucira Health’s “All-In-One” test kit, which on [November 17] became the first rapid, at-home test authorized by the Food and Drug Administration.
The California biotechnology company’s single-use home test kit, which it expects to sell for less than $50, requires a prescription from a doctor. As winter approaches and infections have surged in most states, the at-home test will allow people who are possibly infected to not have to go into clinics or hospitals to get tested.
Unlike rapid antigen tests, which experts warn can be unreliable, the kit will test genetic material in a method similar to the laboratory tests that have become the standard for detecting the virus. After swirling the nasal specimen into a solution, home-testers plug the vial into a portable, battery-operated device, which uses a light to indicate the test result within 30 minutes. A positive test result can be generated in as few as 11 minutes.
The test is expected to be available to patients of Northern California’s Sutter Health and South Florida’s Cleveland Clinic “in the near future” but not reach the national market until early spring 2021.