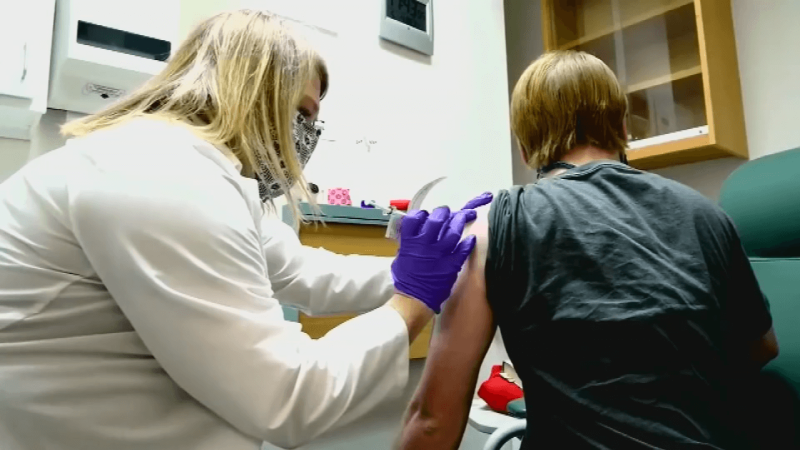
Two criteria for a good vaccine are “effective” and “safe.” The coronavirus vaccine technically named BNT162b2 and developed by industry giant Pfizer and BioNTech meets both standards, according to the professionals who have developed the vaccine, conducted randomized clinical trials and reviewed resulting data during the past several months.
It has received emergency authorization from the FDA and from regulators in the United Kingdom, Canada, Bahrain, Mexico and Saudi Arabia.
Data from the randomized Pfizer-BioNTech trial showed the two-dose vaccine to be 100 percent effective in preventing severe illness from covid-19.
But in roughly half the people who get the shot, it can produce modest side effects, including fever, headache, fatigue and pain at the injection site. That’s typical for most vaccines.
This is not a flaw or a failure, vaccine experts hasten to point out. Side effects are a sign the immune system is kicking into gear, as intended. They’re a feature and not a bug, to borrow the language of computer programmers.
“Things like fever or soreness at the injection site are normal, and actually they indicate that your body is reacting to the vaccine, which is what you want,” said Ellen F. Foxman, an immunologist at the Yale School of Medicine. “That’s a good thing.”