Check out the Genetic Literacy Project’s Global Gene Editing Regulation Tracker and Index
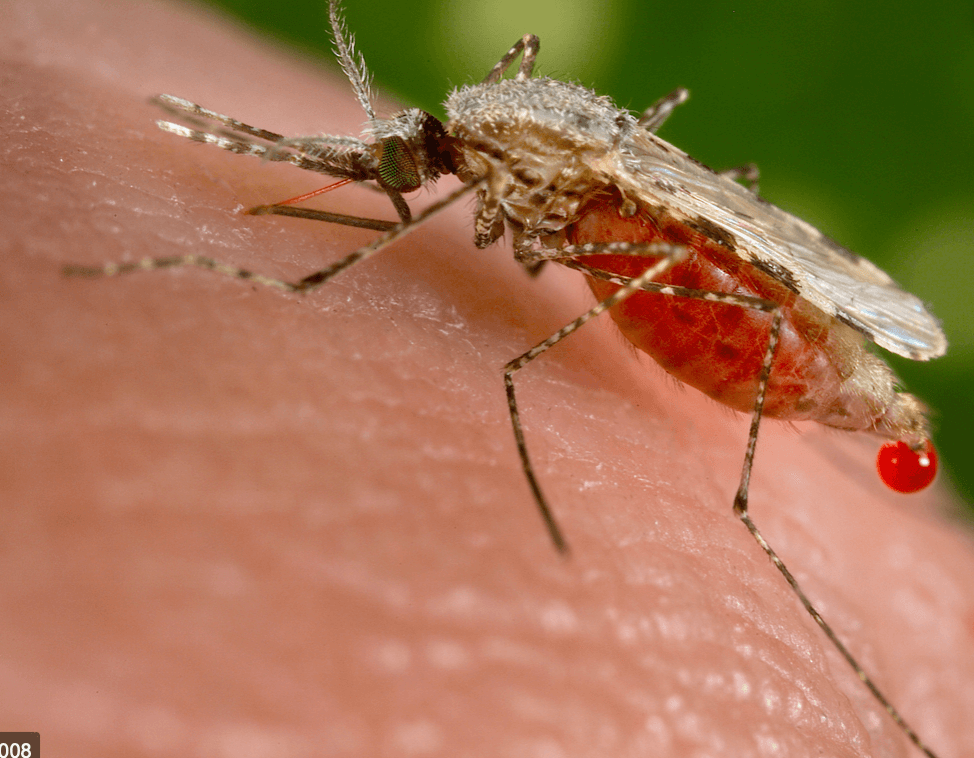
Dengue fever is flying below the global health radar as the fearsome coronavirus outbreak arises out of China. Dengue is an endemic disease in Paraguay, with cases peaking in the summer months when the transmitting Aedes aegypti mosquito proliferates. More than 10,000 suspected cases have been identified in the first weeks of 2020. Dengue fever infects close to 400 million people globally every year.
 What can be done about it? The World Health Organization says the incidence of dengue, which causes high fever and severe muscle, bone and joint pain, has been growing rapidly in recent decades. There is no specific treatment, although a vaccine, untested and unlikely to be widely effective, has been in development for years. So, hope seems distant.
What can be done about it? The World Health Organization says the incidence of dengue, which causes high fever and severe muscle, bone and joint pain, has been growing rapidly in recent decades. There is no specific treatment, although a vaccine, untested and unlikely to be widely effective, has been in development for years. So, hope seems distant.
Stop the presses. There is a promising solution…if politics do not get in the way. It involves in part in deploying an emerging technology: CRISPR gene editing. Scientists have developed, in the laboratory, what’s known as a gene drive, a new biotechnology technique based on naturally occurring phenomenon that can effectively stop mosquitoes from spreading the disease. Using a combination of transgenic and gene editing technology, scientists can prevent the spread of dengue, Zika, malaria and a host of other mosquito-carrying diseases by engineering mosquitoes to replace the disease-carriers and ultimately self-destruct, thus knocking out the insect-borne disease.
But this revolutionary technology, proven in the laboratory and in limited field trials, won’t be helping Benitez or millions of others threatened by disease-carrying pests. Why? Because Paraguay, like most countries, does not have a regulatory structure in place to swiftly capitalize on one of the great, paradigm-shifting, life-saving technologies of our time, in part because anti-biotechnology environmental groups have aggressively campaigned against it.
It’s courting “the end of Nature,” says Canada’s ETC group, one of dozens of NGOs lobbying politicians to ban even research experiments in this area.
No question, as with any new technology, this innovation poses certain risks. But they have to be balanced against it’s life-saving reality. Most scientists believe gene drive technology should be cautiously embraced, and could be the difference between life and death in controlling certain scourges, such as Zika and even serious outbreaks of dengue.
This is just one of dozens of instances in which cutting-edge gene-editing technologies—whether involving gene drives and pest control; CRISPR innovation to improve the sustainability of agriculture; or techniques to snip out diseases embedded in the human genome—are being retarded or blocked by politicians fearful of embracing the future.
GLP tool tracks gene editing regulations worldwide
New technologies deserve a fair hearing. They should thrive or be restricted based on merit, not misinformation. With massive sustainability problems facing our planet, this is no time for advocacy group hackery or industry apologias. With that guiding our work, the Genetic Literacy Project has developed two interactive tools that track and index gene editing and gene drive regulations worldwide, helping to illuminate how regulations can encourage or hinder innovation.
The Global Gene Editing Regulation Tracker and Index summarizes gene editing regulations in agriculture, medicine and gene drives country-by-country, clearly lays out each country’s regulatory timeline, and notes which products and therapies are in development. The tracker also monitors attempts by various advocacy NGOs to block these innovations, and scientists and public interest groups pushing back to give this technology a chance.
![]()
The tracker includes a companion tool, the Gene Editing Regulatory Index, which the GLP developed in conjunction with the Europe-based Consumer Choice Centre. It turns the information in the Regulation Tracker into a quantifiable index that allows for an informative comparison among countries, showing which countries are being more or less conservative in setting up flexible regulatory structures to exploit this revolutionary technology.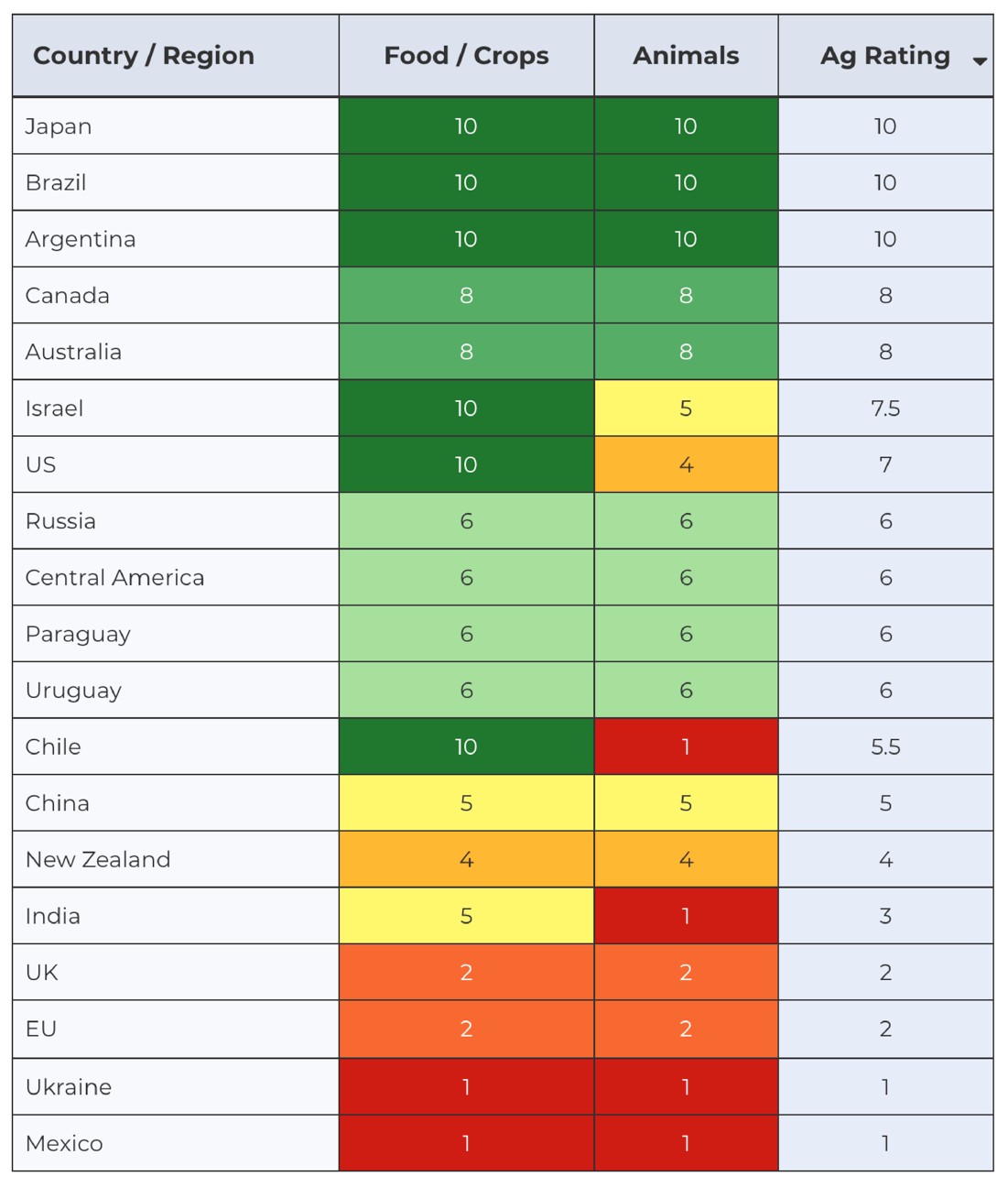
Why is this important?
Gene editing is a breakthrough biotechnology that allows scientists to make precise changes to human, animal and plant DNA. It can be applied to agricultural sustainability, drugs and therapies and industrial uses, and can be combined with other genetic modification techniques to eradicate pests through a new tool called a gene drive.
But these techniques are controversial—scientifically and ethically. To what degree will nations deploy these revolutionary new tools? To what extent does gene editing pose unique hazards that require higher levels of ethical scrutiny and religious debate?
New regulations could help thread the needle of innovation or retard it. They could force countries to openly engage the ethical challenges posed by this emerging technology; they could add unnecessary burdens of time and money, dramatically limiting the benefits of gene editing; or they could help facilitate the cautious but deliberate adoption of countless revolutionary products and treatments.
We’ve seen what happens when fears fanned by special interest advocacy groups take control of the public debate. The GMO revolution that began in the 1990s was set back decades and its vast potential still has not been fully exploited because of misinformation spread by anti-technology advocacy groups. The same advocacy groups that successfully blocked GMOs in Europe, Africa and elsewhere for 20 years are trying to scuttle or indefinitely delay this latest biotechnology breakthrough—which is even more revolutionary.
Gene editing in agriculture
Fears promulgated by special interest groups are already retarding gene editing innovation. Europe is shaping up as an agricultural gene editing backwater. In 2018, the European Court of Justice drew on the legislation passed in 2001 to oversee transgenic crops and decided that gene-edited plants should be regulated the same way as GMOs, which have been effectively blocked since 2001 when politicians capitulated to public fears and advocacy group lobbying to heavily restrict innovation. This political decision came over the objections of Europe’s unified science community.
Its consequences are shaping up as dire. Today, Europe is becoming an agricultural biotechnology backwater. It only has a handful of genetically engineered crops authorized for cultivation and a very cumbersome process for importing GE crops, used mostly for animal feed, from other countries. The latest decision is already resulting in an exodus of research programs to countries with more flexible regulations.
Contrast the EU’s decision to restrictively regulate all gene-edited organisms as GMOs with the regulatory approach in Argentina, which in 2015 became the first (and still only country) to pass new regulations to specifically regulate gene-edited crops as conventional plants as long as they do not contain any DNA from other species.
While Argentina, Japan, Brazil, Colombia, Chile and Australia have all decided to liberally regulate both gene-edited crops and animals, the United States has taken a different approach with gene-edited animals. Unlike crops, the US decided to classify gene-edited animals as drugs, based on regulations developed in the 1930s, which require a much stricter approval process and has slowed research and development in this area.
Gene editing and climate change
Gene editing offers many possible solutions to ecological challenges linked to increased climate variation, especially as the population increases and the planet warms. Drought- and salt-tolerant soybean, heat-tolerant tomato, drought-tolerant maize and salt-tolerant rice have been developed to feed a population-expanding planet and prepare for periods of extreme weather. Another new technology, vertical farms, where plants are grown indoors in stacked layers and often without soil, are now becoming efficient enough to begin to help feed the world’s growing population, providing one way to address the increasing impact of climate change. But scientists argue that for vertical farms to be successful, they will require gene-edited seeds. Gene editing could allow plants to be optimized for vertical farming, including how well they are able to grow vertically and how much water and soil they require.
Low-income countries will be the most devastated by hunger and malnutrition if food production does not increase proportionally. Orphan crops, fruits and vegetables that local farmers rely on for food and income, are especially vulnerable to disease, pests and changes in climate because large agricultural companies tend to spend less time and money on these than staple crops that get bought and sold internationally. Scientists are using gene editing tools to develop cassava, a crucial food in many African countries, that is resistant to the devastating brown streak virus, which is difficult to detect and can lead to complete crop loss. Gene editing would give local farmers access to improved orphan crops, so they do not have to rely on multinational agricultural corporations with large research and development budgets. But, countries in Africa have not yet decided how gene editing will be regulated, and pressure from advocacy groups based in Europe that are virulently anti-GMO and anti-gene editing is fierce.
Human health applications of gene editing
Gene editing is being used to develop a variety of life-saving gene therapies, including Car-T cell therapy, in which a patient’s own immune cells are engineered to fight their cancer. The Global Gene Editing Regulation Tracker and Gene Editing Regulatory Index show that the US and EU are leading the way in research and development of gene and stem cell therapies, with multiple approvals granted over the last few years for cancer treatments, neuromuscular disease, blindness and other diseases.
 In an effort to become an international hub for gene therapy, Japan introduced a controversial accelerated approval system for regenerative medicines, including gene therapy and stem cell treatments, which requires only minimal safety and efficacy data. The UK stands out as the only country yet to officially approve mitochondrial replacement therapy (MRT), popularly known as ‘three-parent IVF’, which was developed to help women avoid having a baby with certain mitochondrial genetic diseases.
In an effort to become an international hub for gene therapy, Japan introduced a controversial accelerated approval system for regenerative medicines, including gene therapy and stem cell treatments, which requires only minimal safety and efficacy data. The UK stands out as the only country yet to officially approve mitochondrial replacement therapy (MRT), popularly known as ‘three-parent IVF’, which was developed to help women avoid having a baby with certain mitochondrial genetic diseases.
Most people are comfortable with the use of gene editing to help treat and cure disease. However, ethical and religious concerns explode when it comes to editing embryos to make genetic changes that would be passed to future generations. Parents could be in position to ensure that their children do not inherit a genetic illness…but germline gene editing can also theoretically be used to make changes to an embryo’s genes for less life-threatening issues, such as hair color, height, athletic ability or intelligence.
A researcher from China sparked international controversy in 2018 when he gene edited human embryos that were brought to term. Since then, China has drafted new, stricter regulations on gene editing. Japan, Russia, and the UK are the only countries that have decided to strictly regulate, but not ban, germline gene editing.
Controlling pests and invasive species with gene drives
Regulations are the most in flux and undecided for an application of gene editing called gene drives. Gene drives are a powerful tool that have the ability to change or eliminate entire species. They are being studied to help eradicate disease-carrying insects and control invasive species. For example, they can be used to develop mosquitoes that only have female offspring. If released into the wild, these mosquitoes would breed with wild malaria-carrying mosquitoes and over time would eliminate the population and therefore stop the spread of malaria.
Gene drives are also being developed to combat dengue fever, invasive species, and even antibiotic resistance. Use of prescription antibiotics and in animal food production have led to an increase in antibiotic resistance, which has become a serious health threat. More than 35,000 people die each year in the US from antibiotic-resistant infections. In 2019, US researchers developed a gene drive that combats antibiotic resistance in E. coli by inactivating an antibiotic resistance gene and creating a positive feedback loop to ensure all cells are affected. More research and tests need to be done, but eventual human applications could include treatments for patients suffering from chronic bacterial infections.

However, genuine scientific concerns have been raised about whether gene drives should be used and how to ensure they are safe and effective. Scientists are still in the process of developing complementary tools, like a kill switch and external overrides to control gene drives once they are released. Consequently, countries worldwide are still debating how they should be regulated. In addition, most gene drives are being developed using transgenic technology (GMOs) that contain foreign genes, as well as gene editing, including CRISPR (synthetic gene drives), which do not, complicating regulatory oversight as gene editing and GMOs are often regulated differently.
Gene drives are being researched in Africa, the US, Australia and New Zealand, but regulations are still not in place. These countries are currently regulating gene drives as GMOs, but only Australia has specifically included gene drives in updated regulations on gene technology.
Where does the GLP stand?
The GLP does not exist to take stands on gene editing or any scientific issue. First, we are not a monolith; we are made up of a tiny core staff and dozens of contributing scientists and journalists. Everyone has their own perspectives on complex issues, especially one as scientifically and ethically freighted as gene editing. Our commitment is to encourage an open playing field in which new technologies are given a fair chance in the global marketplace. We stand for transparency.
Gene editing, as with any evolving technology, should be vigorously scientifically vetted, and open to ethical scrutiny. But like all innovations, it should also not be overly restricted with regulations based on fear rather than accepted scientific facts.
We hope the Global Gene Editing Regulation Tracker and Gene Editing Regulatory Index will encourage science-based scrutiny and advocacy.
The GLP plans to update these tools regularly, as many gene editing regulations are still being developed and implemented.
Kayleen Schreiber, neuroscientist and science communicator, is GLP infographics and data visualization specialist, and director of the gene editing tracker and index
Jon Entine is the executive director of the GLP. Twitter handle: @jonentine